Middle meningeal artery embolization to treat progressive epidural hematoma: a case report
Article information
Abstract
Progressive epidural hematoma is a form of acute epidural hematoma that gradually expands from a small initial hematoma; in cases that are clinically aggravated due to the presence of a mental illness or neurological condition, patients should be surgically treated for evacuation of the hematoma, but poorer outcomes are expected if the patient has several medical co-morbidities for surgery. We experienced two cases of progressive epidural hematoma which were successfully managed by endovascular treatment: an 85-year-old male with medical co-morbidities and a 51-year-old female with a poor-grade subarachnoid hemorrhage resulting from the rupture of a dissecting aneurysm of the vertebral artery. In both cases, a middle meningeal artery embolization was performed and contrast leakage was observed and controlled using cerebral angiography, halting the progression of their epidural hematomas. Thus, endovascular embolization of a middle meningeal artery may play a useful role in salvage therapy in certain complicated situations that limit treatment of the hematoma by surgical evacuation.
Epidural hematomas (EDH) form in the space between the dura mater and the inner surface of the skull. EDH occurs in 10.6% of head trauma patients, mostly as a result of overlying skull fractures,16) and results most often from arterial bleeding from a branch of the middle meningeal artery (MMA).9) A good prognosis is expected in cases with immediate surgical intervention,3)16) while the morbidity and mortality associated with EDH typically result from delayed diagnosis and treatment. The mortality rate of acute EDH ranges from 9–33%, but when the hematoma is evacuated prior to a loss of consciousness, the mortality rate is nearly 0%.16)
Progressive EDH is a form of acute EDH in which a small initial hemorrhage expands over time.3) Progressive EDHs clinically aggravated by the presence of mental disorders or neurological conditions should, ideally, be surgically treated,9) but for patients with several problematic risk factors such as advanced age, poor general physical condition, concomitant underlying diseases, treatment with antithrombotic medications, or in patients with other accompanying emergent problems like subarachnoid hemorrhage (SAH), surgical operation could be fatal in cases involving progressive enlargement of the EDH.8)16) We report two cases of patients with progressive EDH, successfully managed by endovascular treatment.
CASE 1
An 85-year-old male patient was admitted to our hospital via the emergency room after having fallen from a two-meter-high ladder at work; initial brain computed tomography (CT) showed a small EDH in the left temporoparietal area, along with a longitudinal skull fracture (Fig. 1A, B). The patient had a habit of drinking alcohol and suffered from underlying hypertension, diabetes mellitus, and alcoholic liver disease. His initial Glasgow Coma Scale score was 13 (3–4–6), accompanied by general malaise. His mental state continued to deteriorate as he became progressively more confused over time, and his platelet count decreased from 155,000 to 7,000 within a few hours. Follow-up brain CT showed progressive EDH with a subdural hematoma in the same area (Fig. 1C). If the EDH continued to increase in size, a poor outcome was anticipated even following surgical intervention. Because of his advanced age, daily alcohol consumption, and ongoing thrombocytopenia, to stop the expansion of the EDH, we decided to perform the MMA embolization using an endovascular approach.
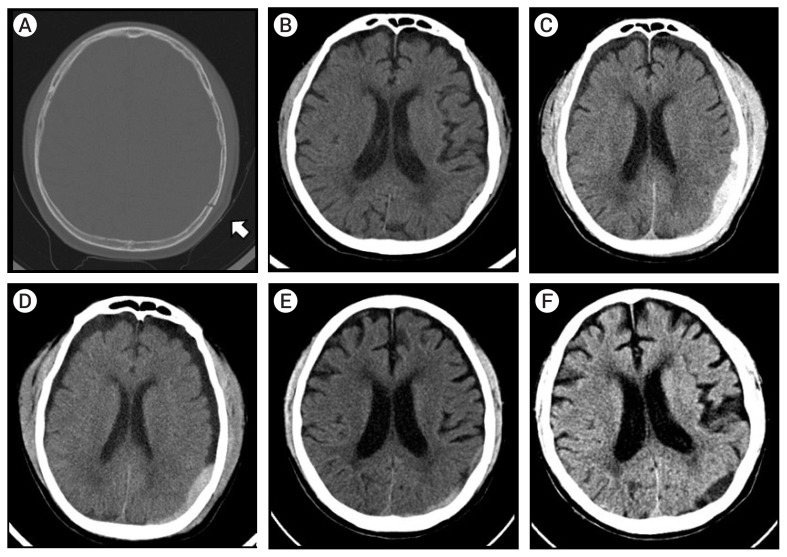
(A) Initial brain computed tomography (CT) showing a left parietal linear skull fracture (white arrow). (B) Initial brain CT with minimal epidural hematoma (EDH) in a fractured site. (C) Pre-procedure brain CT showing increased EDH over time. (D) Three days post-procedure. (E) Brain CT performed two weeks post-procedure showing decreasing amount of EDH (F) Brain CT taken at a one month follow-up; the EDH is almost completely resolved.
Under local anesthesia, the patient was placed in a supine position, followed by shaving and skin preparation of the right inguinal area. electro cardiogram monitoring, oximetry and O2 inhalation were measured throughout the procedure.
The right femoral artery was punctured and a 6-Fr Guider soft tip guiding catheter with a valve (6-Fr, Boston Scientific Corporation, Maple Grove, MN, USA) was inserted by a wire, and was positioned in the left common carotid artery. A solution containing 3,000 U of heparin per 1,000 mL of 0.9% normal physiological saline was infused, and left internal carotid artery (ICA) angiography was performed; critical collateral circulation between the ICA and the external carotid artery (ECA) was ruled out,2) then an Excelsior SL-18 microcatheter (Target Therapeutics, Fremont, CA, USA) was navigated to the MMA. Multiple contrast-leakage points were observed in the parietal branch of the left MMA. MMA embolization was performed, without superselective angiography in the left parietal branch of the MMA, with 45–150 μm Contour polyvinyl alcohol (PVA) embolization particles (Boston Scientific Corporation, Marlborough, MA, USA) (Fig. 2).

(A, B) Preoperative selective angiography of parietal branch of middle meningeal artery (MMA) with contrast leakage (black arrow). (C) Preoperative angiography of the external cerebral artery (ECA) showing contrast leakage (white arrow). (D) Postoperative ECA angiography showing resolved contrast leakage in the parietal branch of the MMA.
Postoperative follow-up
A follow-up brain CT showed no further progression of the EDH (Fig. 1D, E), even though the patient’s platelet count was only 75,000. One week later, the patient was mentally alert without any neurologic deficits or complications, so he was discharged. The EDH was almost resolved one month later, as determined by a follow-up outpatient CT (Fig. 1F).
CASE 2
Presenting illness
A 51-year-old, comatose female patient was transferred to our ER three days after undergoing a hysterectomy due to a uterine myoma performed in another hospital. During the postoperative period, she complained about the sudden onset of bursting headache; brain CT showed spontaneous SAH with intraventricular hemorrhage (IVH) (modified Fischer grade 4). To make the situation worse, the patient experienced cardiac arrest during the brain CT, and return of spontaneous circulation (ROSC) was achieved only after six minutes of cardiopulmonary resuscitation (CPR). The patient was transferred to our hospital immediately after ROSC.
The subsequent CT-angiography performed in our hospital revealed showed a dissecting aneurysm of the vertebral artery (VA) on the right side. Due to acute hydrocephalus, emergent external ventricular drainage (EVD) was performed to reduce intracranial pressure (ICP). The procedure was done in the CT room under real-time CT-assisted guidance. Cerebrospinal fluid (CSF) leakage occurred following ventricular puncture, along with a small degree of acute EDH at the puncture site (Fig. 3A). The degree of EDH progressively increased on a one minute follow-up brain CT scan and another twist drill trephination was performed at the hematoma site to drain the EDH (Fig. 3B). The patient was then moved to the angiography room for the endovascular procedure.
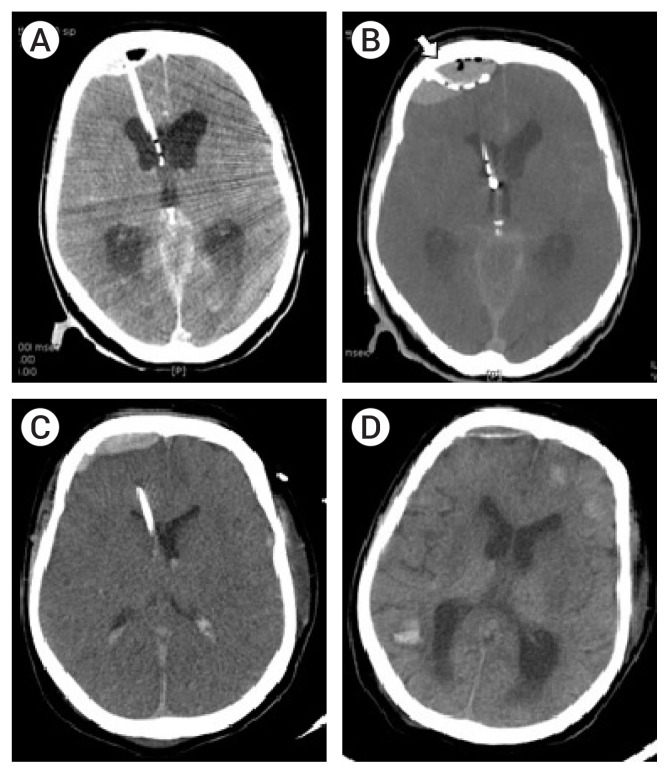
(A) Post-external ventricular drainage (EVD) CT scan showing small epidural hematoma (EDH) at the EVD site. (B) Additional burr hole made lateral to the first burr hole (white arrow). The brain CT shows increasing size of the EDH with pneumocephalus. (C) Two days after EVD, the catheter to treat the EDH is removed. (D) Follow-up brain CT in one month after the procedure. The EDH is almost completely resolved, but brain CT shows multiple intracerebral hemorrhages due to thrombocytopenia.
Endovascular procedure and clinical findings
Under general endotracheal anesthesia, a6-Fr Chaperone guide catheter (MicroVention, Tustin, CA, USA) with a valve was inserted by a wire and was positioned in the left common carotid artery (CCA).
To prevent expansion of the EDH in the right frontal area, right MMA embolization was attempted first. Right ICA angiography was performed and critical collateral circulation between the ICA and ECA was ruled out. An Excelsior SL-18 microcatheter (Stryker Neurovascular, Fremont, CA, USA) was navigated to the right MMA. A contrast-leakage point was observed in the frontal branch of the right MMA. The right MMA was occluded completely with 45–150 μm Contour PVA embolization particles without complications (Fig. 4A, B).
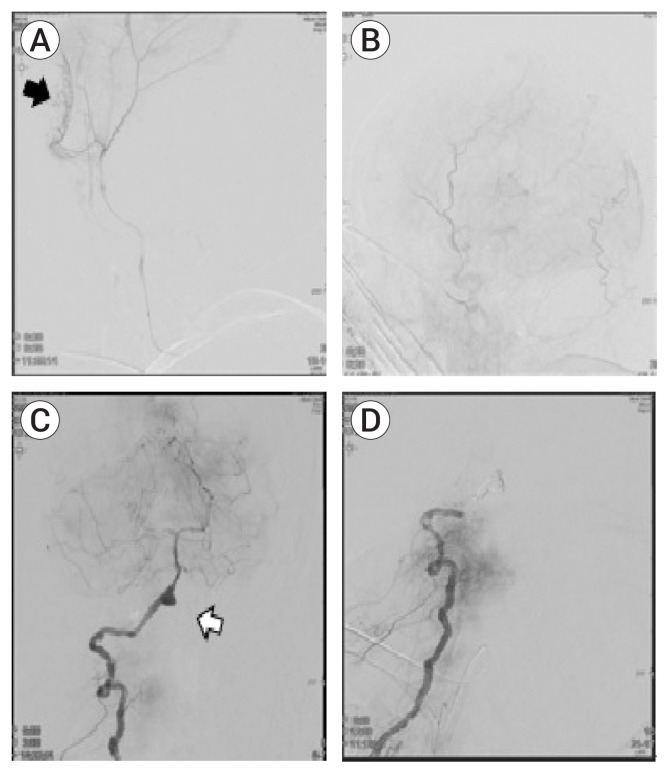
(A) Selective angiography of the MMA showing a contrast leakage site (black arrow). (B) Postoperative angiography of the ECA showing resolution of the contrast-leakage site. (C) Preoperative right vertebra artery (VA) angiography showing an aneurysm in the V4 segment (white arrow). (D) Angiography after trapping of the right VA with coil. MMA, middle meningeal artery; ECA, external carotid artery.
After embolization of the right MMA, endovascular trapping of the right VA dissecting aneurysm was performed and was finished successfully with preservation of the right PICA (Fig. 4C, D). During the procedure, drainage of the EDH and CSF through EVD was monitored carefully. A follow-up CT after the procedure revealed no further progression of the EDH, and the catheter was removed two days after the operation (Fig. 3C).
The remaining EDH was almost completely resolved three weeks post-surgery (Fig. 3D), as determine by a follow-up CT, but the patient’s neurological condition did not improve due to the initial hypoxic damage that occurred during CPR. Two months post-ictus, the patient passed away due to medical complications.
DISCUSSION
The MMA is a clinically meaningful structure for treating various diseases, such as chronic subdural hematoma (SDH), meningioma, arteriovenous malformation or fistula, indirect revascularization to treat moyamoya disease, and even migraine.17) These conditions could be effectively treated by endovascular or surgical approaches via the MMA.6)17)
For recurrent chronic SDHs after burr hole trephination or spontaneous SDHs due to medical problems such as tumors, blood dyscrasias, or liver cirrhosis, an MMA embolization could be attempted as an alternative treatment for recurrence of chronic SDH, based on the fact that the feeding artery to the outer membrane in chronic SDH originates in the MMA.4–6)12)15)17)
Progressive EDH is typically observed over the course of repeated CT scans, which can reveal a new EDH or an increase in EDH size compared to the initial CT findings.3) The incidence of progressive EDH ranges from 5.6% to 13.3%, and the proportion of EDHs treated surgically ranges from 5.5% to 32%.2) In the case of progressive expansion of an EDH with clinical deterioration, urgent surgical evacuation is required to protect the brain from secondary injuries, such as those resulting from cerebral edema or ischemia. The most common cause of acute EDH is an injury to the MMA, and MMA ligation could be a therapeutic option to prevent EDH progression.
The endovascular treatment of acute EDH was first described by Suzuki et al. in 2004;14) to date, there have been many advances in endovascular treatment.2)9)14) Recently, Peres et al. demonstrated the safety and efficacy of the procedure for treating patients with small EDHs. Successful embolization was achieved without surgical intervention, resulting in the eventual disappearance of the hematoma,9)11) but the role of endovascular treatment of acute EDH has not been well established in neurosurgical fields, and Bortoluzzi et al. have described the necessity for determining proper clinical criteria, collecting imaging data, and identifying the optimal timing for performing endovascular treatment of acute EDH.2)
In our study, two cases of progressive EDH were successfully managed with endovascular treatment. In the first case, the patient was an elderly male with a hepatic problem due to heavy drinking, with underlying hypertension and diabetes mellitus. According to other literature,10)13) the level of operative risk of the patient was moderate degree The size of the EDH increased over time to a point where the operative outcome was predicted to be poor due to his medical deterioration and neurological condition.
In the second case, the EDH occurred in an elderly female patient during an emergent EVD procedures. The size of the EDH increased rapidly, as observed in a one-minute-interval CT scan, and endovascular management was deemed necessary for treating the poor-grade spontaneous SAH due to a VA dissecting aneurysm. It was a situation of wavering between EDH evacuation and endovascular trapping of the VA dissecting aneurysm as an initial treatment. Eventually both cases of progressive EDH with complicating factors, such as high surgical risk or possible fatality necessitating urgent treatment, were successfully managed by endovascular MMA embolization.
The endovascular management of small EDHs has been proposed as a safe and effective treatment option1)7)11)14) and as an alternative to avoid surgical evacuation.9) Furthermore, in the case of progressive EDHs which initially started out as small to moderate in size and did not require evacuation, MMA embolization increases the likelihood of a good prognosis by halting the enlargement of the EDH in complicated cases.
CONCLUSIONS
Progressive EDH is a form of acute EDH in which there is a continuous expansion of the hematoma, requiring surgical intervention in cases of clinical or radiological deterioration., Endovascular management of progressive EDH through MMA embolization may be an effective treatment option for certain patients at high surgical risk or heightened possibility of fatal complications instead of surgical evacuation. And could be performed in cases with the patients who reveal active bleeding points from the angiogram.
Notes
Disclosure
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
