 |
 |
| J Cerebrovasc Endovasc Neurosurg > Volume 24(2); 2022 > Article |
|
Abstract
The hypoglossal canal (HC) is an unusual location of the posterior fossa dural arteriovenous fistula (AVF), which usually occurs in the transverse or sigmoid sinus. Herein, we report a case of HC dural AVF successfully treated with transvenous coil embolization using detachable coils in a 68-year-old woman who presented with headache and left pulsatile tinnitus for 2 months. Brain magnetic resonance imaging (MRI) and cerebral angiography revealed left HC dural AVF. The pulsatile bruit disappeared immediately after the procedure. Follow-up MRI showed complete disappearance of the fistula. Precise localization of the fistula through careful consideration of the anatomy and transvenous coil embolization using a detachable coil can facilitate the treatment for HC dural AVF.
Intracranial dural arteriovenous fistulas (AVFs) are defined as abnormal connections between the meningeal arteries and dural sinuses or veins within the dura mater. Although most posterior fossa dural AVFs usually occur in the transverse or sigmoid sinus, the hypoglossal canal (HC) is an unusual location of the posterior fossa dural AVF. To date, only a few case series of HC dural AVFs have been reported [3,6,9]. Unusual location and similar appearance on angiography might result in misdiagnosis as marginal or inferior petrosal sinus dural AVF [1,5,7]. HC dural AVFs are usually fed by the meningeal branches of the bilateral ascending pharyngeal artery via the HC, occipital artery via the mastoid foramen, and vertebral artery via the foramen magnum. As the anterior condylar vein within the HC has numerous anastomoses, symptoms and treatment modalities for HC dural AVF depend on the venous drainage pattern [6,8]. Here, we describe a case of a patient with HC dural AVF treated with transvenous coil embolization using detachable coils.
A 68-year-old woman presented with left pulse-synchronous tinnitus and headache for 2 months. Physical examination demonstrated an audible bruit over the left mastoid process. Bruit decreased during the ipsilateral carotid artery compression. Ocular symptoms, such as exophthalmos or chemosis, were not observed. Magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA) were performed to rule out the vascular origin. MRA demonstrated abnormal signal hyperintensity around the HC (the “magic wand appearance”) (Fig. 1A, 1B). Computed tomography (CT) of the bone showed bone erosion and left HC expansion (Fig. 1C). Transfemoral cerebral angiography revealed a dural arteriovenous fistula (AVF) in the anterior condylar vein within the HC, fed by the neuromeningeal branches of the bilateral-ascending pharyngeal artery, occipital artery (Fig. 2A, 2B, 2C), and vertebral artery (Fig. 2D). The dural AVF drained into the internal jugular vein, inferior petrosal sinus, and vertebral venous plexus. Although retrograde flow drained into the cavernous sinus through inferior petrosal sinus, cortical reflux was not observed (Cognard type IIa). Schematic illustration of the anatomy of dural AVF is described in Fig. 2E. One week after cerebral angiography, transvenous embolization was performed under general anesthesia to occlude the venous pouch of the anterior condylar vein. After positioning the 5-Fr angiocatheter in the left external carotid artery and a 6-Fr guiding catheter in the left internal jugular vein, an Excelsior 1018 microcatheter was inserted into the anterior condylar vein via the jugular bulb. The precise location of the distal tip of the microcatheter was confirmed using microcatheter angiography. The dilated venous pouch was packed from the point that connected the vertebral venous plexus to the inferior petrosal sinus using three detachable coils (Fig. 3A). Although the venous pouch was not completely packed due to microcatheter kickback after detaching the last coil, delayed angiography after 15 min revealed the complete disappearance of the shunt flow. However, contrast stagnation remained in the mural channel surrounding the HC (Fig. 3B, 3C). CT scan after embolization confirmed the presence of a coil mass in the left HC (Fig. 3D). The pulse-synchronous tinnitus and bruit completely disappeared immediately after the procedure. The patient was managed with steroids for the prevention of hypoglossal palsy and was discharged without neurologic deficits. At 6 months after the procedure, follow-up MRA showed complete disappearance of the dural AVF (Fig. 4).
Since the concept of anterior condylar dural AVF within the HC was first described by Ernst et al. in 1999 [5], it has been considered a different subset of dural AVFs from the marginal sinus or inferior petrosal sinus dural AVF. Manabe et al. [6] advocated the expression “HC dural AVF” as an ideal term rather than “dural AVF of the anterior condylar vein” after confirming coils packed in the expanded HC in nine patients.
It is important to understand the venous system anatomy in the posterior fossa and vertebral venous system. The inferior petrosal sinus was connected to the jugular bulb after the branching of the anterior condylar vein, which passes through the HC and connects the jugular vein to the vertebral venous plexus. Numerous connections of the anterior condylar vein with the surrounding venous structures may have several different venous drainage patterns. Symptoms and signs of hypoglossal dural AVF depend on the drainage pattern. Although most patients with hypoglossal dural AVF usually present with tinnitus and headache, stenosis development or anterograde drainage route obstruction can result in reflux into the cavernous sinus or perimedullary vein. In these patients, aggressive symptoms, such as ophthalmic symptoms or myelopathy, may develop.
The transvenous approach and coil embolization of venous structures are considered the most effective treatment modalities for hypoglossal dural AVF [3,6]. Transarterial embolization with embolic material can be considered as an alternative or complementary modality in patients where a venous approach is not possible. However, embolic material poses a potential risk of cranial nerve palsy and recurrence. For successful transvenous embolization of a hypoglossal dural AVF, careful consideration of the fistular anatomy can facilitate the navigation and precise location of the microcatheter. The extent and location of the fistula are best delineated on contralateral carotid arteriography. The extent of fistular occlusion must be delineated before transvenous coil embolization because incomplete occlusion with venous outflow obstruction might change the venous drainage pattern and cause venous hypertension. Unexpected compromise of the adjacent inferior petrosal sinus during transvenous coil embolization should be avoided. The precise location of the microcatheter in the dilated venous pouch should be confirmed with superselective angiography and three-dimensional angiography. During coil embolization, microcatheter kickback is a major concern. Detachable coils are more easily controllable and cause less microcatheter kickback compared with pushable coils. According to Manabe et al., stiffer coils packed in the HC induce compression of the hypoglossal nerve and subsequent palsy [2,4,6]. Softer coils, such as detachable coils, may help prevent hypoglossal palsy. Despite the effort to completely occlude the venous pouch, unexpected kickback of the microcatheter and incomplete occlusion can occur. However, delayed thrombus formation and complete occlusion might be expected if the coil frame covers the entire venous pouch.
We described the case of a patient with HC dural AVF treated with transvenous coil embolization using detachable coils. Careful consideration of the fistular anatomy and precise location of the microcatheter is important. Our case shows that transvenous coil embolization using detachable coils is an effective treatment modality for the treatment of HC dural AVF. Overpacking with stiffer coils should be avoided to prevent postoperative hypoglossal palsy.
Fig. 1.
Magnetic resonance angiography shows abnormal signal hyperintensity around the left hypoglossal canal (the “magic wand appearance”) (arrow) (A, B). A bone imaging computed tomography shows bone erosion and expansion of left hypoglossal canal (arrow) (C).
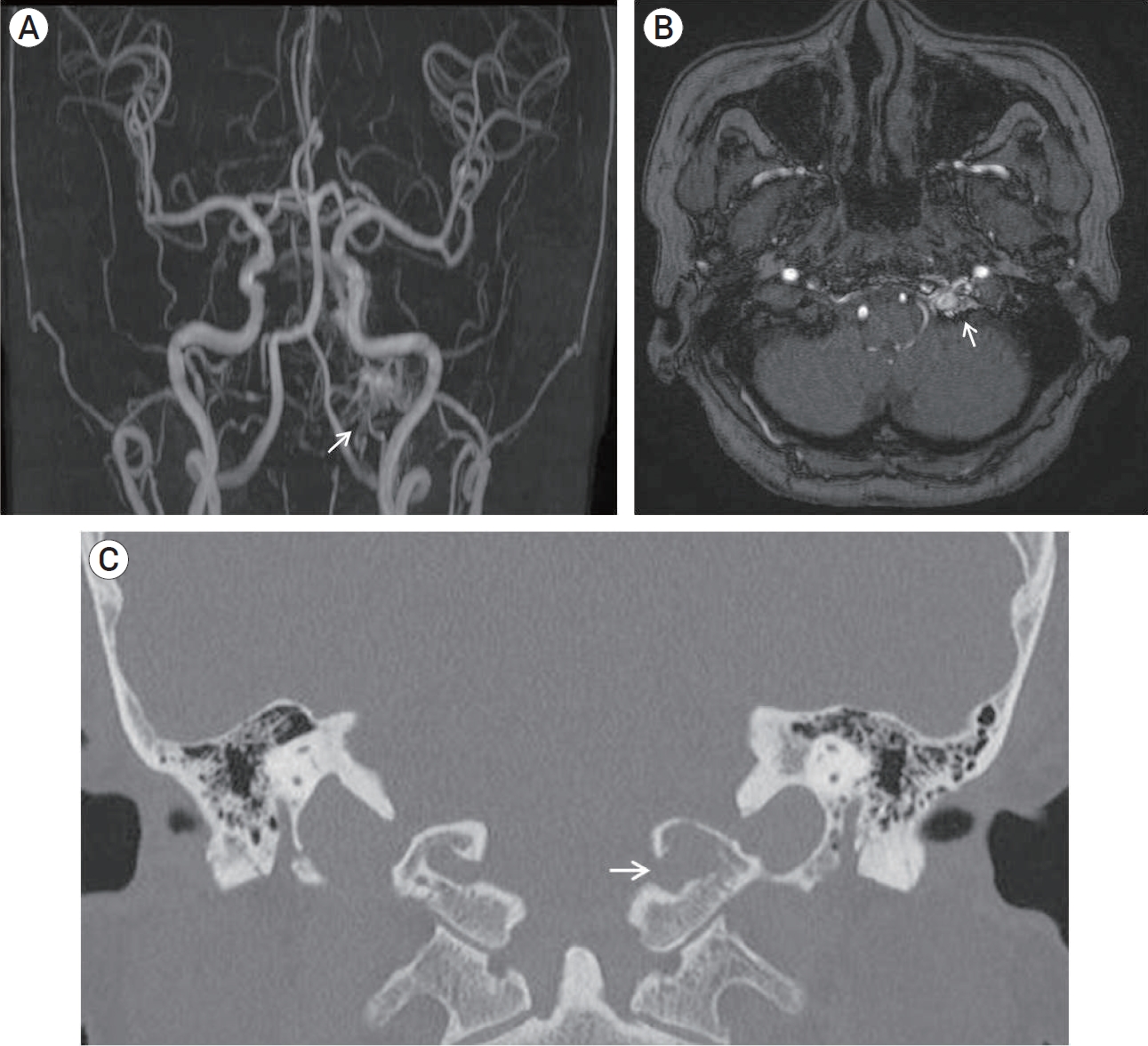
Fig. 2.
Cerebral angiography reveals a dural arteriovenous fistula in the anterior condylar vein within the left hypoglossal canal (HC) fed by neuromeningeal branches of the bilatreral ascending pharyngeal artery (APA), occipital artery (A, B, C) and vertebral artery (D), which drained into internal jugular vein (IJV), cavernous sinus (CS) through inferior petrosal sinus (IPS), and vertebral venous plexus (VVP). Schematic illustration shows detailed anatomy of dural arteriovenous fistula (E).
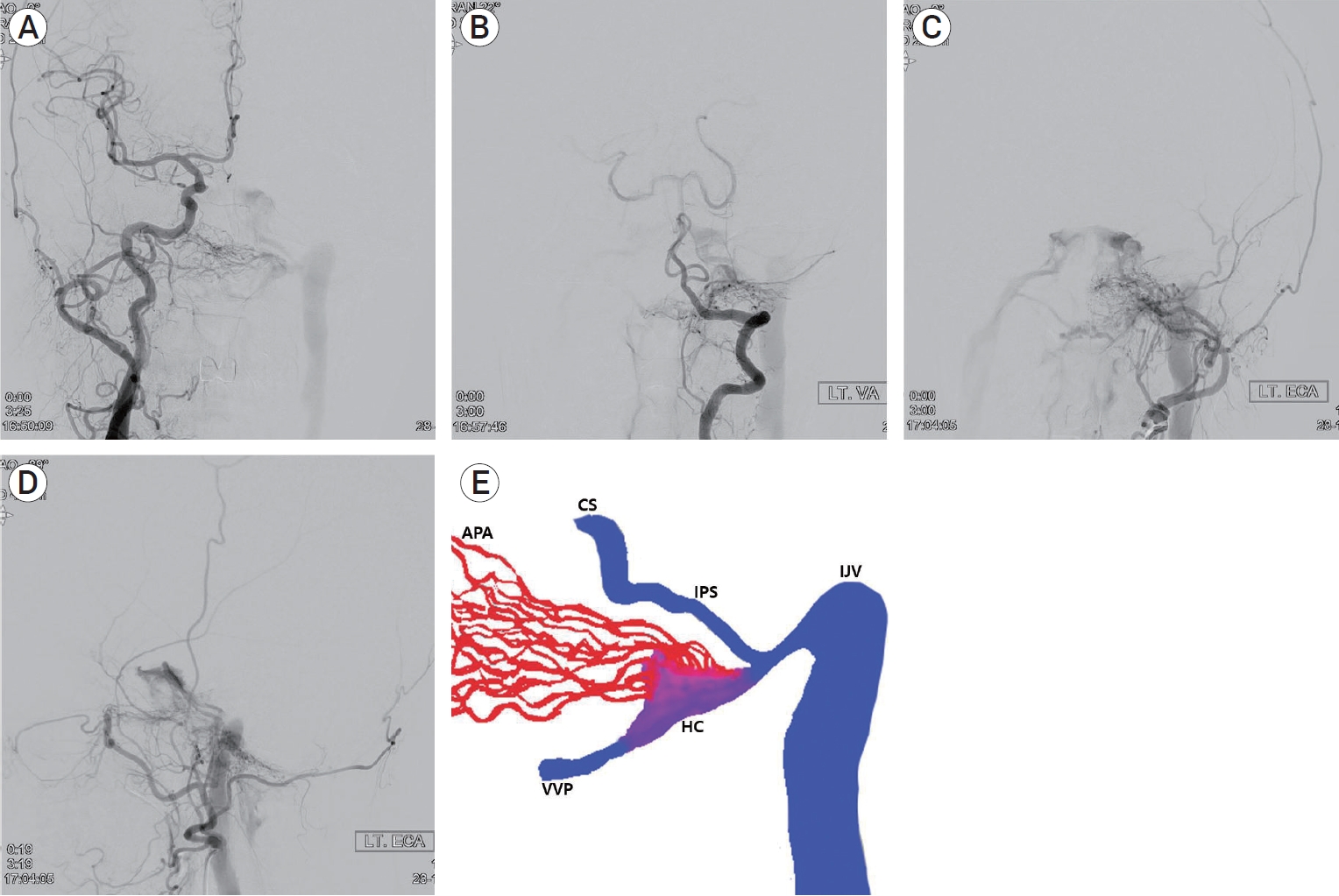
Fig. 3.
Microcatheter angiography demonstrated dilated venous pouch connected with vertebral venous plexus (arrow) and inferior petrosal sinus (arrowhead) (A). Final angiography showed complete occlusion of dural arteriovenous fistula and contrast stagnation around the hypoglossal canal (arrow) (B, C). CT shows coil mass in the hypoglossal canal (D). CT, computed tomography.

REFERENCES
1. Barnwell SL, Halbach VV, Dowd CF, Higashida RT, Hieshima GB. Dural arteriovenous fistulas involving the inferior petrosal sinus: angiographic findings in six patients. AJNR Am J Neuroradiol. 1990 May;11(3):511-6.


2. Blomquist MH, Barr JD, Hurst RW. Isolated unilateral hypoglossal neuropathy caused by dural arteriovenous fistula. AJNR Am J Neuroradiol. 1998 May;19(5):951-3.


3. Choi JW, Kim BM, Kim DJ, Kim DI, Suh SH, Shin N-Y, et al. Hypoglossal canal dural arteriovenous fistula: incidence and the relationship between symptoms and drainage pattern. J Neurosurg. 2013 Oct;119(4):955-60.


4. Combarros O, Alvarez de Arcaya A, Berciano J. Isolated unilateral hypoglossal nerve palsy: nine cases. J Neurol. 1998 Feb;245(2):98-100.


5. Ernst R, Bulas R, Tomsick T, van Loveren H, Aziz KA. Three cases of dural arteriovenous fistula of the anterior condylar vein within the hypoglossal canal. AJNR Am J Neuroradiol. 1999 Nov-Dec;20(10):2016-20.


6. Manabe S, Satoh K, Matsubara S, Satomi J, Hanaoka M, Nagahiro S. Characteristics, diagnosis and treatment of hypoglossal canal dural arteriovenous fistula: report of nine cases. Neuroradiology. 2008 Aug;50(8):715-21.


7. McDougall CG, Halbach VV, Dowd CF, Higashida RT, Larsen DW, Hieshima GB. Dural arteriovenous fistulas of the marginal sinus. AJNR Am J Neuroradiol. 1997 Sep;18(8):1565-72.



-
METRICS

-
- 1 Crossref
- 0 Scopus
- 2,917 View
- 69 Download
- ORCID iDs
-
Jun Hyong Ahn

https://orcid.org/0000-0002-8529-6757 - Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



