 |
 |
| J Cerebrovasc Endovasc Neurosurg > Volume 24(4); 2022 > Article |
|
Abstract
Development of Internal Carotid Artery pseudoaneurysms (ICAp) after transsphenoidal surgery is extremely rare, occurring only in 0.4% of cases. Surgical treatment of ICAp poses a real challenge to the neurosurgeon as treatment may require parent vessel sacrifice or artery reconstruction with bypass grafting. Furthermore, surgical resolution of these lesions is rarely reported in the literature.
The internal carotid artery is prone to iatrogenic injury in transsphenoidal surgery due to its frequent involvement in pituitary adenomas. Intracranial pseudoaneurysms may be at high risk for rupture and increased morbidity and mortality. Here we present a case of a patient with an ICAp rupture two months after transsphenoidal surgery for a pituitary adenoma.
The coexistence of cerebral aneurysms and pituitary adenomas is rare, with an estimated incidence of approximately 3.6%. Intracranial aneurysms and pituitary adenomas have been reported by multiple authors in multiple studies, yet a clear association between them still does not exist. Cases of infectious and traumatic aneurysms in the context of pituitary tumors have been reported [4].
Traumatic intracranial aneurysms are rare and are classified in direct or indirect according to Burton et al. Direct trauma occurs during penetrating head injury or surgery, whereas indirect trauma results from blunt head injury [5].
Wilson and Dempsey reported one of the first traumatic aneurysms during a transsphenoidal surgery in their series of 250 pituitary adenoma surgeries [14]. Despite being infrequent, iatrogenic pseudoaneurysm following endoscopic endonasal pituitary surgery is the most common vascular complication and damage to the cavernous ICA is the most common type of vascular injury after transsphenoidal surgery [9]. Intracranial pseudoaneurysms are uncommon and among the most difficult lesions to treat with surgery or endovascular means without parent artery sacrifice. Treatments include a wide variety of microsurgical and endovascular techniques such as clipping, trapping (with or without bypass), carotid artery ligation, balloon occlusion, coil embolization, stent-assisted coil embolization, covered stent placement, and the use of Onyx [11].
A 39-year-old man presented to our institution after presenting with tonic-clonic seizures and neurological deterioration that required advanced airway management. Clinical history was relevant for a giant non-functioning pituitary adenoma which was treated via transsphenoidal surgery two months ago (Fig. 1). Patient did not have a computed tomography (CT) angiogram or magnetic resonance angiography (MRA) performed before or after trans sphenoidal surgery. Postoperative course was uneventful. Neurological examination was carried out under deep sedation and was only relevant for reactive mydriasis. Brainstem reflexes were preserved.
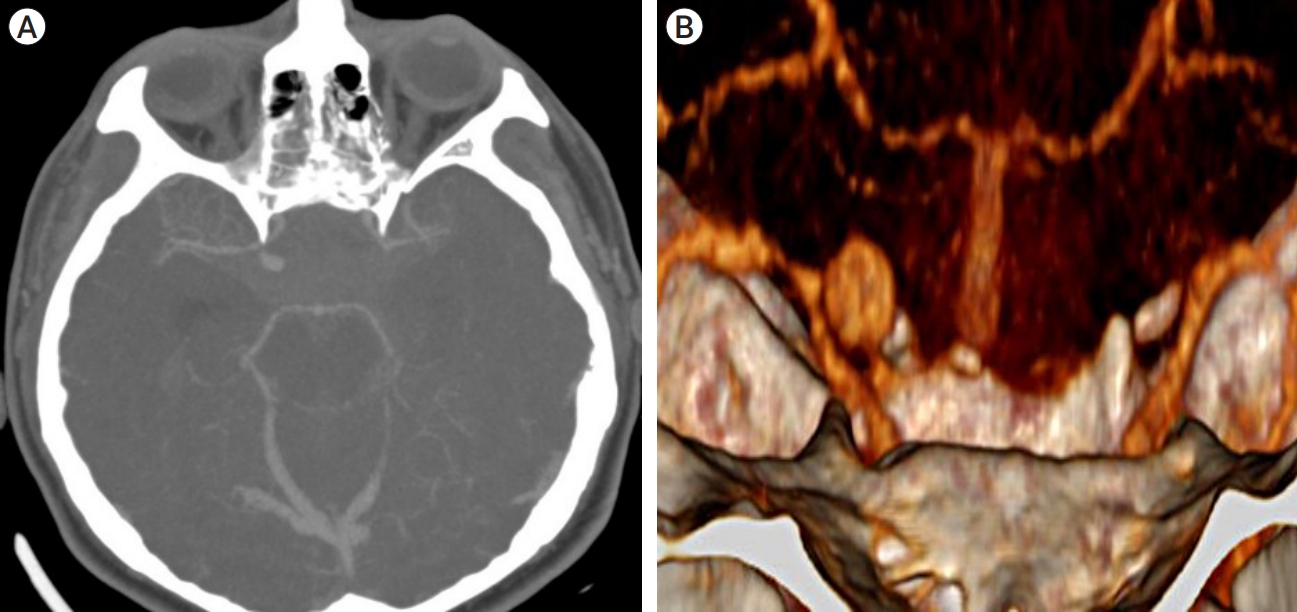
The patient underwent a simple CT which revealed Fisher IV subarachnoid hemorrhage and hydrocephalus (Fig. 2). Furthermore, an extensive subacute stroke in the left anterior cerebral artery territory was noticed. A cerebral angiotomography was performed and showed a saccular aneurysm of the right internal carotid artery involving the communicating segment. The aneurysm had a neck of 8 mm and measured 7.1×5.5 mm. The dome of the aneurysm was oriented medially. Anterior cerebral arteries were not identified, suggesting severe vasospasm (Fig. 3). Due to the patient’s severe clinical status transfemoral catheter cerebral angiography was not performed.
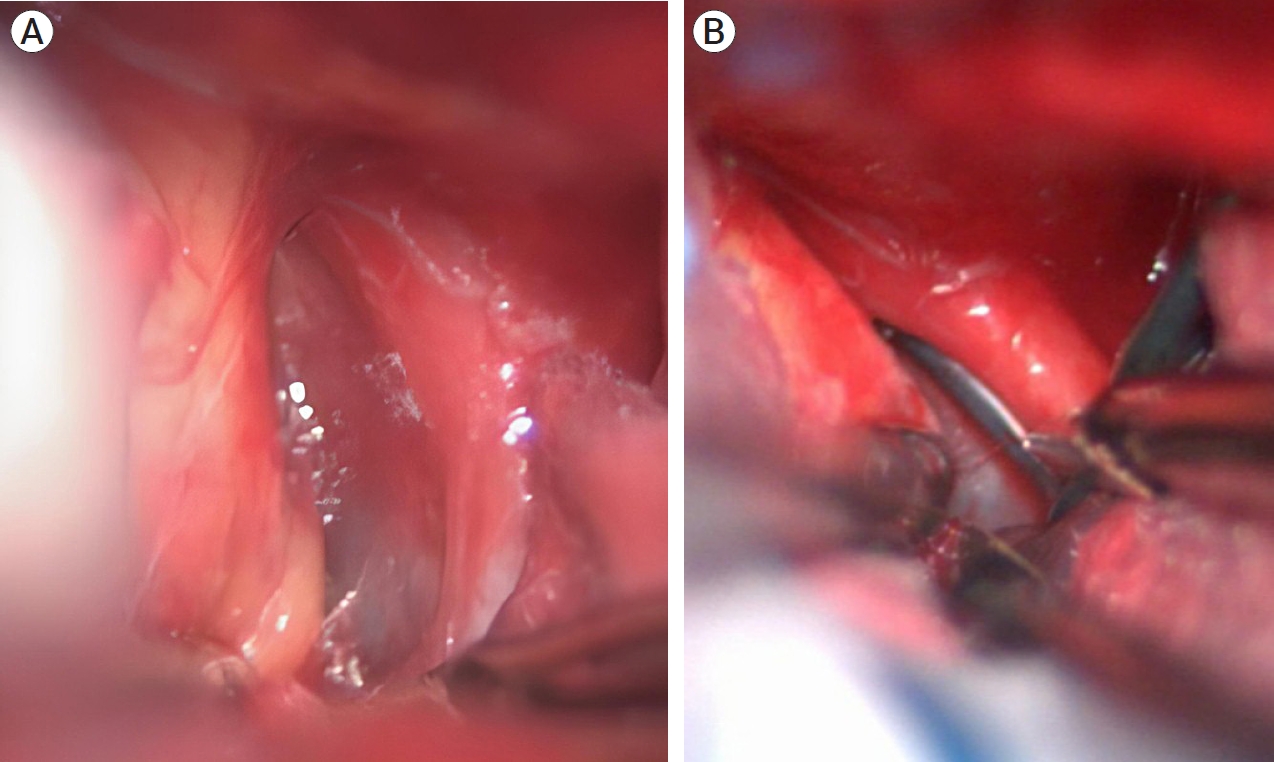
Due to the severe cerebral edema, incipient malignant stroke and hydrocephalus, surgical management was chosen, as the need for decompressive craniectomy for patient´s survival was foreseen. First, a left precoronal ventriculostomy was placed to treat hydrocephalus and achieve brain relaxation for better surgical manipulation. A conventional right pterional craniectomy and surgical clipping of the aneurysm via subfrontal approach was performed (Fig. 4). Clinoidectomy was not necessary for adequate aneurysm clipping, however, temporary cervical carotid control was achieved using a vascular clamp due to the high risk of intraoperative rupture. Aneurysm clipping was accomplished successfully by placing a 6 mm fenestrated clip that included the ipsilateral communicating segment of the ICA in the clip’s fenestration. Postoperative cerebral angiotomography revealed adequate aneurysm exclusion with distal flow preservation and extensive subacute stroke in middle cerebral artery and anterior cerebral artery (Fig. 5).
Postoperative course took place in the Intensive Care Unit. Patient underwent tracheostomy and gastrostomy and weaning from ventilation was possible two weeks after surgery. Afterwards the patient received floor care and was discharged successfully.
Intracranial pseudoaneurysms are uncommon and among the most difficult lesions to diagnose and treat. Iatrogenic pseudoaneurysm may occur after intracranial procedures such as tumor resection, stereotactic biopsy and transsphenoidal surgery. ICA pseudoaneurysms resulting from iatrogenic injuries during transsphenoidal surgery (TSS) are not true aneurysms, as they lack a true, full-thickness wall and are covered only by a fragile connective tissue layer [7].
The reported incidence of ICA injuries during TSS for pituitary tumors ranges between 0.55 and 1.1% [6,11,15]. ICA pseudoaneurysms resulting from iatrogenic injuries during TSS are similar to blood blister-type aneurysms histologically (indicating focal artery laceration), and geographically (occurring at non-branching sites of the ICA), with the possibility for rapid enlargement and rupture with a 30-50% reported mortality rate [1,10-12]. Although shrinkage and spontaneous resolution of intracranial pseudoaneurysms have been reported, clinical series have reported rupture rates of up to 60% prior to definitive treatment [2,11].
Pseudoaneurysms may result in complications such as subarachnoid hemorrhage, epistaxis, and carotid cavernous fistula with resultant death or permanent neurologic deficit [8]. Subarachnoid hemorrhage after transsphenoidal surgery for pituitary adenoma is an extremely rare occurrence with only three case reports in the English literature, and hemorrhage can occur in the first days or even months after surgery [11].
Risk factor associated with vascular injury during transsphenoidal surgery includes anatomic variations of the sphenoid bones, an abnormally short distance between ICAs, ICA displacement by the lesion, tumor invasion of the cavernous sinus, tumor adhesion to the ICA, acromegaly with distortion of the local anatomy, and unusual tortuosities in the ICA. Previous surgery, previous external radiotherapy, chemoradiotherapy, or prior treatment with bromocriptine for pituitary tumors have also been reported [2].
Routine preoperative cerebral MRA is highly recommended because patients treated for pituitary adenoma are likely to develop intracranial arterial aneurysms as iatrogenic complications. Furthermore, in untreated patients, the incidence of coincidental cerebral aneurysm is higher than would be expected by chance alone. If excessive bleeding occurs during or after transsphenoidal surgery, even after successful intraoperative tamponade, immediate vascular imaging should be obtained to rule out vessel wall damage [13].
Various treatment strategies have been used in the management of pseudoaneurysms, including observation. Nevertheless, endovascular embolization is the treatment of choice and the choice of endovascular technique is based on the location of the pseudoaneurysm, vascular anatomy and patient’s clinical status. Treatment modalities include coiling, liquid embolization, stent assisted coiling, covered stent implantation and flow diverting stents [16].
Surgical intervention is mandatory when endovascular treatment has failed or when significant mass effect and edema is present in ruptured cerebral pseudoaneurysms [13]. Surgical options include direct clipping, suturing, wrapping-clipping, ligation of the parent artery, and trapping with or without bypass [16]. Because of the absence of real neck and sac wall, which involve an extremely high risk of perioperative rupture, proximal vascular control is mandatory in the treatment of pseudoaneurysms. There are several methods for obtaining proximal control including endovascular (temporary balloon occlusion), surgical (dissection of the ICA in the neck, temporary clipping of parent vessel) and even pharmacological approaches (transient cardiac arrest induced by adenosine. There is no standardized method for accomplishing proximal vascular control and choosing a specific method depends on the surgeon’s experience and resources availability [3].
Occurrence of subarachnoid hemorrhage months after transsphenoidal surgery is extremely rare and its surgical management is not well documented in medical literature.
Endovascular therapy is the treatment of choice when possible, as it has been the most studied therapy in these cases. Nevertheless, ruptured pseudoaneurysms that cause significant edema or mass effect and/or are located in distal intracranial branches have a much better prognosis when surgical treatment is chosen in this context. Furthermore, preoperative routine evaluation with MRA is imperative since the incidence of coincidental pituitary adenoma and cerebral aneurysm is higher than would be expected by chance alone.
Fig. 1.
(A) Preoperative cranial MRI in sagittal and coronal T1-weighted sections. (B) A giant sellar tumor consistent with pituitary adenoma with heterogeneous gadolinium enhancement. MRI, magnetic resonance imaging
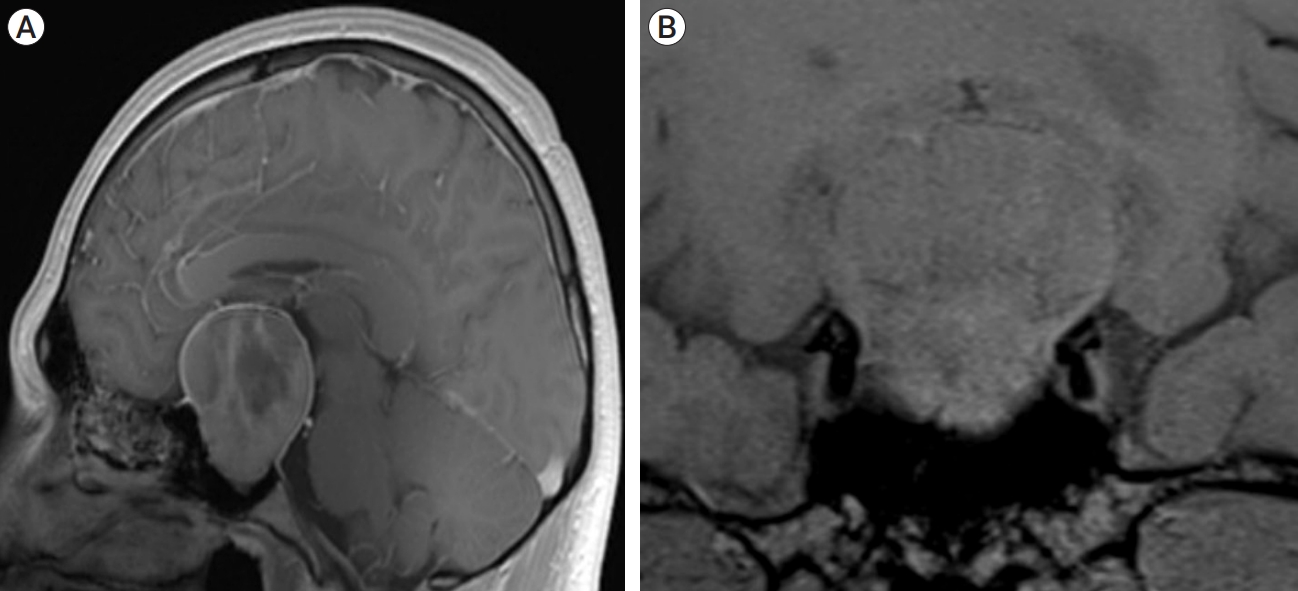
Fig. 2.
(A) Preoperative cranial CT scans in axial sections demonstrate Fisher IV subarachnoid hemorrhage and hydrocephalus. (B) Sagittal view demonstrates an extensive anterior cerebral artery stroke that prompted surgical resolution of the case. CT, computed tomography

Fig. 3.
(A) Cerebral angiotomography with 3D reconstruction, demonstrating the right internal carotid artery pseudo aneurysm and the dome’s medial orientation (B) as well as its proximity to the sellar surgical region suggesting pseudoaneurysm.
REFERENCES
1. Alzhrani G, Sivakumar W, Park MS, Taussky P, Couldwell WT. Delayed complications after transsphenoidal surgery for pituitary adenomas. World Neurosurg. 2018 Jan;109:233-41.


2. Amenta PS, Starke RM, Jabbour PM, Tjoumakaris SI, Gonzalez LF, Rosenwasser RH. Successful treatment of a traumatic carotid pseudoaneurysm with the Pipeline stent: case report and review of the literature. Surg Neurol Int. 2012 3:160.



3. Andrade-Barazarte H, Luostarinen T, Goehre F, Kivelev J, Jahromi BR, Ludtka C, et al. Transient cardiac arrest induced by adenosine: a tool for contralateral clipping of internal carotid artery-ophtalmic segment aneurysms. World Neurosurg. 2015 Dec;84(6):1933-40.

4. Bulsara KR, Karavadia SS, Powers CJ, Paullus WC. Association between pituitary adenomas and intracranial aneurysms: an illustrative case and review of the literature. Neurol India. 2007 Oct-Dec;55(4):410-2.


5. Burton C, Velasco F, Dorman J. Traumatic aneurysm of a peripheral cerebral artery. Review and case report. J Neurosurg. 1968 May;28(5):468-74.

6. Ciceri EFM, Regna-Gladin C, Erbetta A, Chiapparini L, Nappini S, Savoiardo M, et al. Iatrogenic intracranial pseudoaneurysms: neuroradiological and therapeutical considerations, including endovascular options. Neurol Sci. 2006 Nov;27(5):317-22.



7. Goyal N, Basheer N, Suri A, Mahapatra AK. Subarachnoid hemorrhage after transsphenoidal surgery for pituitary adenoma: a case report and review of literature. Neurol India. May-Jun. 2012 60(3):337-8.
8. Karadag A, Kinali B, Ugur O, Oran I, Middlebrooks EH, Senoglu M. A case of pseudoaneurysm of the internal carotid artery following endoscopic endonasal pituitary surgery: endovascular treatment with flow-diverting stent implantation. Acta Medica (Hradec Kralove). 2017 60(2):89-92.


10. Medel R, Crowley RW, Hamilton DK, Dumont AS. Endovascular obliteration of an intracranial pseudoaneurysm: the utility of Onyx. J Neurosurg Pediatr. 2009 Nov;4(5):445-8.


11. Nariai Y, Kawamura Y, Takigawa T, Hyodo A, Suzuki K. Pipeline embolization for an iatrogenic intracranial internal carotid artery pseudoaneurysm after transsphenoidal pituitary tumor surgery: case report and review of the literature. Interv Neuroradiol. 2020 Feb;26(1):74-82.




12. Nerva JD, Morton RP, Levitt MR, Osbun JW, Ferreira MJ, Ghodke BV, et al. Pipeline embolization device as primary treatment for blister aneurysms and iatrogenic pseudoaneurysms of the internal carotid artery. J Neurointerv Surg. 2015 Mar;7(3):210-6.


13. Raymond J, Hardy J, Czepko R, Roy D. Arterial injuries in transsphenoidal surgery for pituitary adenoma; the role of angiography and endovascular treatment. AJNR Am J Neuroradiol. 1997 Apr;18(4):655-65.


14. Wilson CB, Dempsey LC. Transsphenoidal microsurgical removal of 250 pituitary adenomas. J Neurosurg. 1978 Jan;48(1):13-22.


- TOOLS
-
METRICS

-
- 4 Crossref
- 0 Scopus
- 1,877 View
- 48 Download
- ORCID iDs
-
Luis Alfonso Castillejo Adalid

https://orcid.org/0000-0002-8680-4493 - Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



