Primary Angiitis of Central Nervous System related intracranial aneurysm with spontaneous occlusion after immunomodulatory treatment
Article information
Abstract
Primary Angiitis of the Central Nervous System (PACNS) is an uncommon disease with kaleidoscopic clinical manifestations. Ischemic strokes are commoner than their hemorrhagic counterpart. Intracranial pseudoaneurysms are rarely reported in PACNS cohorts. We hereby describe the case of a 39-year-old female, who presented for evaluation of acute onset of left middle cerebral artery (MCA) ischemic stroke, with cerebral angiogram showing multifocal stenosis and irregularities in intracranial blood vessels with an aneurysm arising from the lenticulostriate branch of the left MCA M1 segment. A diagnosis of probable PACNS was made and patient initiated on immunomodulatory treatment with corticosteroids. 12 weeks follow up neuroimaging studies revealed resolution of the previously described intracranial aneurysm, thereby postulating the possibility of a pseudoaneurysm related to the underlying angiitis.
INTRODUCTION
Primary Angiitis of the Central Nervous System (PACNS) is an uncommon disease with kaleidoscopic clinical manifestations [1,6-8]. On account of the broad differentials, a high index of clinical suspicion and extensive multidimensional investigations are essential to arrive at a diagnosis and institute timely management with immunomodulators. The complexity of PACNS is partially contributed by rarity of the condition, with the annual incidence of PACNS estimated to be 2.4 cases per 10,00,000-person years (95% confidence interval 0.3–4.4) [7]. Ischemic strokes are commoner than the hemorrhagic counterpart, and intracranial aneurysms are rarely reported with the postulated pathogenic mechanism of the latter being angiitis [1,6-9]. Our index case is a 39-year-old female who presented for evaluation of an acute ischemic left middle cerebral artery (MCA) stroke, who on further evaluation had multifocal stenoses and irregularities involving intracranial vessels along with an aneurysm arising from the lenticulostriate branch of the left MCA M1 segment. A diagnosis of probable PACNS was made with patient initiated on immunomodulatory therapy, with follow up neuroimaging studies documenting resolution of the intracranial aneurysm. The dynamic resolution of intracranial aneurysm in association with PACNS treatment highlights the importance of presenting our case, which will add to the literature evidence of the kaleidoscopic manifestations of this hitherto rare disease.
CASE DESCRIPTION
A 39-year-old female of middle east origin with no prior medical co-morbidities except for smoking presented with acute onset of right sided weakness. Neurological examination was notable for right sided facial droop with dysarthria, dense flaccid hemiplegia of right upper and lower extremity. Baseline non-contrast computerized tomography (NCCT) Head revealed hypodensities in the left basal ganglia, lentiform nucleus and left periventricular white matter, suggestive of acute infarcts. These findings were confirmed on a non-contrast magnetic resonance imaging (MRI) brain which showed restricted diffusion in the above regions (Fig. 1A, B, C). There was no evidence of prior ischemic/hemorrhagic strokes, additional white matter hyperintensities or cerebral microbleeds. CT Angiogram of the head and neck vessels revealed irregularity and mild stenosis of the left MCA M1/M2 and severe stenosis of the left posterior cerebral artery (PCA) distal P1 segments. In addition, focal dilatation was noted in a prominent lenticulostriate branch arising from left M1 segment suggestive of an aneurysm. There were no other obvious markers for intra or extracranial cerebrovascular atherosclerotic disease. Baseline blood investigations (Table 1) revealed grossly unremarkable complete blood counts, renal and hepatic function. Erythrocyte sedimentation rate, C Reactive Protein, Hba1c and lipid panel are also detailed in Table 1. Transthoracic echocardiography and continuous cardiac telemetry were negative for any cardioembolic source.

(A, B, C) Magnetic resonance imaging (MRI) Brain diffusion-weighted imaging (DWI) sequences reveals hyperintense signal involving the left basal ganglia, internal capsule and periventricular region suggestive of acute infarcts in the left middle cerebral artery (MCA) territory (black arrows). Digital Substraction Angiogram (DSA) shows aneurysm involving the left lenticulostriate aneurysm on coronal sequences of left internal carotid artery injection (D; white arrow), with lateral sequences (E) revealing the same aneurysm (white arrow) and underlying left M1 MCA stenosis. (F) Anteroposterior view of vertebral artery injection shows left P1 posterior cerebral artery (PCA) stenosis (black arrow).
The patient was initiated on acetylsalicylic acid 100 mg daily for secondary stroke prevention. Given high suspicion for PACNS causing multifocal inflammatory stenosis and possibly aneurysm formation, further investigations were pursued. A catheter Digital Substraction Angiogram (DSA) was performed which confirmed the irregular segments associated with stenosis in the left MCA and left PCA. It also clearly delineated a left lenticulostriate aneurysm in the proximal trunk measuring 4 mm in largest diameter (Fig. 1D, E, F). Globular shape of the aneurysm without a neck along with delayed filling and contrast stagnation were suggestive of a pseudoaneurysm. A contrast enhanced MRI (CEMRI) Brain did not show any areas of abnormal leptomeningeal or nodular parenchymal enhancement apart from expected enhancement of the subacute infarct. High resolution contrast enhanced MRI Brain vessel wall imaging (HRVWI) revealed diffuse wall thickening and concentric enhancement in the region of the left MCA and left PCA stenotic segments, along with additional thick nodular enhancement in the area of maximum left P1 stenosis (Fig. 2B, C).
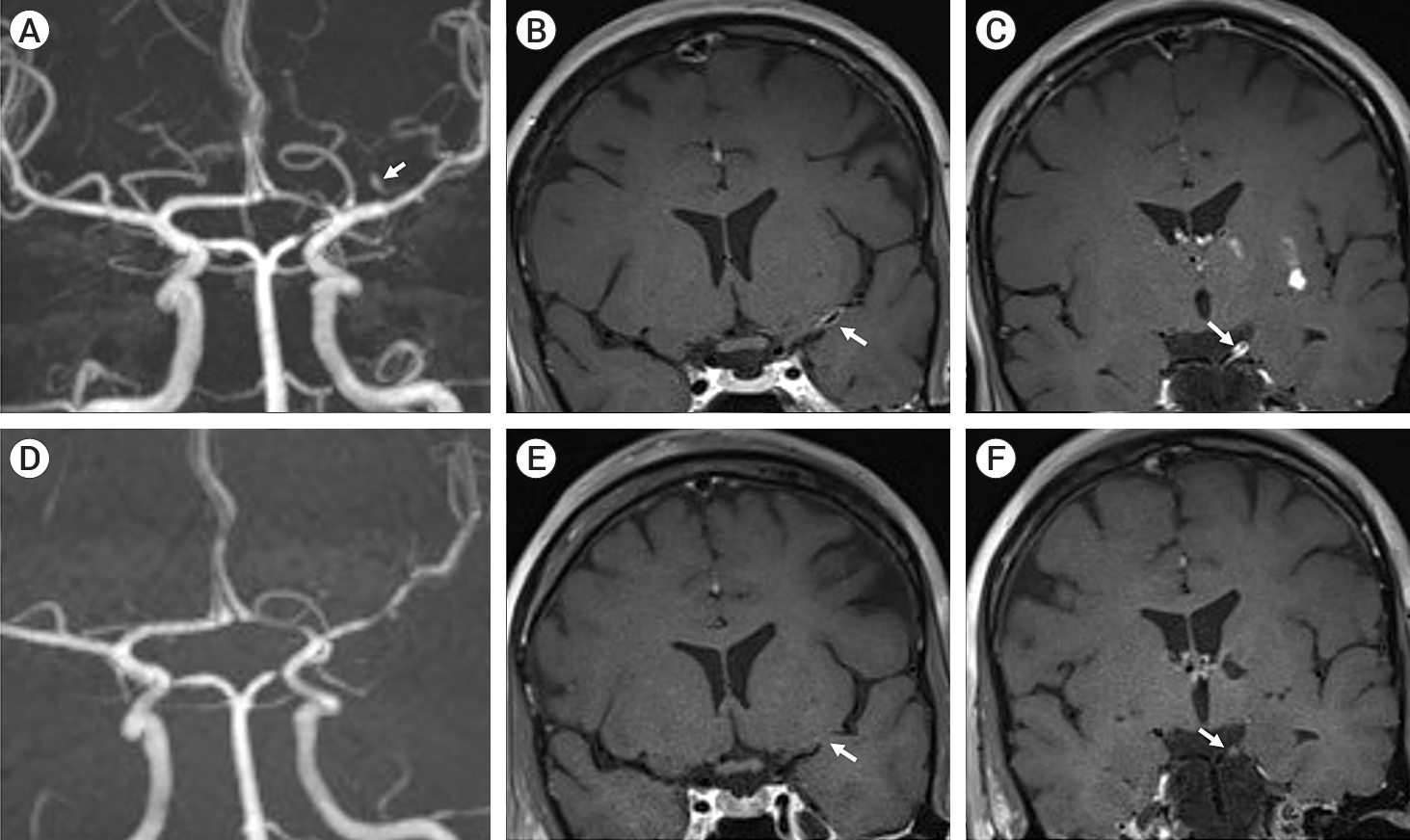
(A) Baseline contrast enhanced magnetic resonance angiogram (MRA) of the cerebral vessels reveals left lenticulostriate aneurysm (white arrow), with underlying left middle cerebral artery (MCA) M1 and P1 posterior cerebral artery (PCA) stenosis. (B) Baseline coronal T1 post contrast sequences of high-resolution MR vessel wall imaging (VWI) study demonstrate concentric wall thickening and circumferential enhancement of the left M1 MCA (B; white arrow) and left PCA P1 segment (C; white arrow) suggestive of vasculitis. (D) Follow up MRA of cerebral vessels reveals disappearance of the aneurysm with underlying stable stenosis of left MCA M1 and PCA P1 segment. Follow up T1 post contrast coronal images of MR VWI depicts resolution of the enhancement involving the left MCA M1 (E; white arrow) and left PCA P1 segment (F; white arrow).
Additional blood investigations (Table 1) included a negative systemic vasculitis, anti-nuclear antibody panel and hypercoagulable panel (including lupus anticoagulant, IgM/IgG anticardiolipin and beta2 glycoprotein antibodies). She underwent lumbar puncture which revealed 16 WBC per mm3 (Normal range 0–5 WBC/mm3), 94% mononuclear with no atypical cells, with high protein – 64 mg/dl (Normal range – 20–40 mg/dl), and normal glucose. Cerebrospinal fluid (CSF) infectious work-up revealed negative meningitis and encephalitis panel, CSF Mycobacterium tuberculosis polymerase chain reaction (PCR) and CSF cultures. Contrast enhanced CT Chest, Abdomen and Pelvis was normal. Ophthalmology evaluation revealed no signs of active retinal vasculitis. On account of the clinical presentation as well as the corroborative neuroimaging and CSF findings, a high possibility of PACNS was considered. A meningo-cortical biopsy (MCB) was discussed with the patient, but risk of false negative result due to sampling bias given lack of additional lesions amenable to biopsy was explained. The patient declined biopsy.
Given the high likelihood of probable large vessel PACNS, she was given a course of intravenous methylprednisolone 1,000 mg daily for five days followed by maintenance of oral prednisolone (60 mg/day). Given lack of definite pathological diagnosis due to unavailability of tissue and non-progressive clinical course, aggressive standard induction with Cyclophosphamide was not pursued. Conservative management without open surgical or endovascular intervention was advised for the lenticulostriate aneurysm given unruptured status and high risk for treatment related complications. She remained clinically stable with no relapses. Corticosteroid tapering was initiated 8 weeks after initiation. Follow up CEMRI Brain twelve weeks after initiation of corticosteroids revealed no new stroke and no areas of abnormal contrast enhancement. Follow up HRVWI revealed significant improvement in the vessel wall enhancement with only mild residual enhancement in both the left MCA and PCA (Fig. 2E, F). The stenosis persisted suggestive of post inflammatory fibrosis. Lastly, the lenticulostriate aneurysm could not be visualized suggestive of spontaneous occlusion (Fig. 2D). The corticosteroids were gradually tapered over 6 months and then discontinued. The patient has remained clinically stable with no relapses at 1 year.
DISCUSSION
Our case highlights an acute clinical and neuroimaging presentation of probable large vessel PACNS with acute ischemic stroke and additional unruptured cerebral aneurysm which spontaneously occluded on immunomodulatory treatment without requiring surgical intervention. The presence of the lenticulostriate aneurysm in close proximity to the diseased M1 segment is likely the result of inflammatory vasculitis affecting the perforator vessels also and subsequently contributing to aneurysm genesis. This along with the spontaneous occlusion of the aneurysm under corticosteroid treatment alone strongly suggests that the aneurysm was related to the angiitis itself.
PACNS is an uncommon and poorly understood inflammatory vasculitis involving only the central nervous system (brain and spinal cord) without an underlying systemic inflammatory disease [1,6-8]. It is more common in males (1.4-2 times) [1,7,8]. with a median age of 50 years (range 17-84 years) [7,8]. PACNS is known for heterogenous and non-specific clinical manifestations, with headaches being the most common (63%) followed by altered cognition (50%) and focal neurological symptoms (44%) [1,7,8]. Ischemic lesions are far more common (50%) in PACNS. Intracranial hemorrhage is much rarer noted only in 10-14% of PACNS study subjects [1,6-8], with intracerebral hemorrhage being the most common type (57-60%) [6,7].
The radiological gold standard investigation for PACNS is cerebral angiography. The most frequently noted abnormality is classical finding of multiple areas of smooth stenosis in different vessel territories with interposed areas of ectasia or normal luminal architecture termed as “beaded” appearance [1,7,8], other findings include vascular occlusions, collateral formation, and prolonged circulation time. Despite haemorrhagic presentations, microaneurysms are rarely seen. The pathophysiology of the aneurysm development related to angiitis is unclear. Many patients with histologically proven PACNS may have an entirely normal angiogram [5]. This is because DSA is limited by low specificity of 30% with sensitivity ranging from 40-90%, partly contributed by the limited utility in cerebral small vessels less than 500 µm in size [7,8].
Therefore targeted MCB is the gold standard investigation for PACNS, with a sensitivity of 53-63%, with granulomatous vasculitis (58%) being the most common type [7,8]. The factors which led on to a diagnosis of PACNS in our index case were demographic profile of the patient (young female with ischemic stroke in the absence of significant vascular risk factors), CSF picture (mononuclear pleocytosis with elevated protein and negative infective markers), vascular imaging studies (with DSA depicting multi-focal stenosis and HRVWI demonstrating diffuse wall thickening and homogenous concentric enhancement of the affected vessel segments) with exclusion of alternate plausible etiologies, despite the lack of MCB.
As alluded to earlier in our case, the imaging characteristics of the aneurysm on DSA were suggestive of a pseudoaneurysm. It is plausible that impaired vascular wall integrity from the angiitis causes a disruption of the entire arterial wall including the adventitia. An encapsulated hematoma forms outside the vessel, being restricted only by perivascular connective tissues but it continues to communicate with the injured artery, thus predisposing it to rebleeding. Overall, intracranial pseudoaneurysms itself are rare and represents about 1% of all intracranial aneurysms with an associated mortality of 20% or higher [9]. The most common cause of pseudoaneurysm is trauma while other causes include iatrogenic, infectious disease, radiation exposure, connective tissue disease, and sometimes spontaneous [9].
Aneurysms caused by inflammatory angiitis are being increasingly recognized in PACNS. Pillai et al. report a case of histopathologic ally proven PACNS initially presenting with ischemic stroke, and while on treatment with corticosteroids and cyclophosphamide developed intraventricular hemorrhage 9 months later [4]. DSA which was initially normal showed significant progression of vasculitic changes, with interval development of multi-vascular luminal irregularities and multiple aneurysms in the left posterior inferior cerebellar artery. Given ruptured status, the patient underwent surgical clipping and wrapping of the aneurysms. Gaillard et al. also described a patient with PACNS who harboured 6 microaneurysm in various vasculatures, that appeared to improve with immunomodulatory treatment alone [3]. In the French cohort ‘COVAC (COhort of patients with primary VAsculitis of the Central nervous system)’, which studied 52 patients with PACNS, 5 patients (13%) were found with intracranial microaneurysms on conventional angiography [2].
Optimal management of aneurysms related to PACNS is unknown given the exquisite rarity of the problem. The knowledge that ruptures of pseudoaneurysms in particular is associated with high rates of morbidity and mortality makes the management even more challenging. For ruptured cases, it would seem prudent to use standard microsurgical and endovascular interventions, on an individualized basis to secure the aneurysm. For unruptured aneurysms, our case may offer support for conservative management with immunosuppressive treatment alone for the angiitis. While the true natural history of pseudoaneurysms is unclear, complete spontaneous occlusion is an uncommon occurrence. This may result from remodelling/ healing of the vascular wall, or thrombosis of the hematoma. If conservative approach is pursued, then close imaging follow-up is advisable to assess interval changes and consider prompt treatment if worsening aneurysmal characteristics develop.
CONCLUSIONS
PACNS is a challenging disease due to its heterogenous clinical presentation and wide differential diagnosis. Our case of an unruptured cerebral aneurysm with spontaneous occlusion adds to the pathology caused by PACNS. Aneurysms related to PACNS are extremely rare, and is plausibly from pseudoaneurysm formation due to the disruption of the arterial wall. While treatment of aneurysms related to PACNS needs to be individualized, foremost based on rupture status, there may be a role for conservative management with immunomodulatory treatment alone in unruptured aneurysms.
Notes
Disclosure
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.

