Imaging follow-up strategy after endovascular treatment of Intracranial aneurysms: A literature review and guideline recommendations
Article information
Abstract
Objective
Endovascular coil embolization is the primary treatment modality for intracranial aneurysms. However, its long-term durability remains of concern, with a considerable proportion of cases requiring aneurysm reopening and retreatment. Therefore, establishing optimal follow-up imaging protocols is necessary to ensure a durable occlusion. This study aimed to develop guidelines for follow-up imaging strategies after endovascular treatment of intracranial aneurysms.
Methods
A committee comprising members of the Korean Neuroendovascular Society and other relevant societies was formed. A literature review and analyses of the major published guidelines were conducted to gather evidence. A panel of 40 experts convened to achieve a consensus on the recommendations using the modified Delphi method.
Results
The panel members reached the following consensus: 1. Schedule the initial follow-up imaging within 3-6 months of treatment. 2. Noninvasive imaging modalities, such as three-dimensional time-of-flight magnetic resonance angiography (MRA) or contrast-enhanced MRA, are alternatives to digital subtraction angiography (DSA) during the first follow-up. 3. Schedule mid-term follow-up imaging at 1, 2, 4, and 6 years after the initial treatment. 4. If noninvasive imaging reveals unstable changes in the treated aneurysms, DSA should be considered. 5. Consider late-term follow-up imaging every 3–5 years for lifelong monitoring of patients with unstable changes or at high risk of recurrence.
Conclusions
The guidelines aim to provide physicians with the information to make informed decisions and provide patients with high-quality care. However, owing to a lack of specific recommendations and scientific data, these guidelines are based on expert consensus and should be considered in conjunction with individual patient characteristics and circumstances.
INTRODUCTION
Preventing rupture (in cases of unruptured aneurysms) or rebleeding (in cases of ruptured aneurysms) is the fundamental goal of aneurysm treatment. In several institutions, endovascular embolization has emerged as the primary treatment modality for intracranial aneurysms [6,17,24]. Additionally, improved endovascular techniques, including balloon-assisted coiling, stent-assisted coiling, flow diversion, and flow disruption, have enabled the treatment of a greater number of aneurysms, including those with complex and challenging anatomical configurations [5,19,21,25,28].
Although endovascular coil embolization has proven effective in clinical practice, its long-term durability remains of concern. After conventional endovascular coiling, approximately 20% of patients who undergo endovascular treatment (EVT) experience a reopening of the aneurysm or neck, and approximately 9% of them require retreatment to maintain long-term protection against hemorrhage [8,18]. Therefore, the treated aneurysms must be followed up over time to ensure a durable occlusion. Moreover, in patients with multiple cerebral aneurysms, untreated small residual aneurysms may grow or transform into shapes that require future intervention; de novo aneurysms not previously observed may also be discovered. According to some studies, these possibilities range from 5 to 10% [13,14].
Given the importance of monitoring patients who receive treatment for intracranial aneurysms, optimal follow-up imaging protocols must be established, especially as life expectancy has increased owing to advancements in medical technology. However, currently, there is no established scientific evidence or guidelines that define the ideal methodology for follow-up imaging of treated aneurysms, including the imaging frequency, duration, and techniques.
The Korean Neuroendovascular Society (KoNES) aimed to address this gap by formulating clinical practice guidelines for the follow-up observation of patients who undergo EVT for cerebral aneurysms. These guidelines describe the current follow-up strategies after EVT for intracranial aneurysms and provide evidence based on a literature search and consensus among physicians who treat intracranial aneurysms in clinical practice. The guidelines aim to provide physicians with information on follow-up strategies after EVT for intracranial aneurysms, enabling them to make informed decisions and provide high-quality care to their patients. Notably, the ultimate decision regarding the follow-up strategy for each patient should be made by the responsible healthcare providers, patients, and/or their caregivers, considering the patients’ specific circumstances. These guidelines do not intend to limit the practice of healthcare professionals or serve as a basis for evaluating insurance claims and should never be used as the sole basis for legal judgments in cases involving medical care provided under specific clinical circumstances.
MATERIALS AND METHODS
The Clinical Practice Guideline Committee of the KoNES was formed by members of the KoNES, as well as appointed members of the Korean Stroke Society, Korean Society of Interventional Neuroradiology, and Korean Society of Cerebrovascular Surgeons, to conduct the literature review and develop the guidelines.
In addition, an expert panel consisting of 40 experts (33 from the KoNES and seven from the Korean Society of Interventional Neuroradiology) was organized to achieve a consensus on the recommendations proposed by the members of the group drafting the guidelines. In April 2023, using the modified Delphi method, the expert panel members reached a consensus, and the Clinical Practice Guidelines Oversight Committee and participating academic societies reviewed and approved the draft prepared by the members who drafted these guidelines.
Evidence searches and data analyses
The group drafting the guidelines of the Clinical Practice Guideline Committee of the KoNES conducted a systematic review of the relevant literature and major published guidelines. This review involved comprehensive searches of various databases, including PubMed, EMBASE, and the Cochrane Library, and the data were collected and analyzed until March 2022. Rather than create new guidelines, the Committee adopted a developmental approach by adapting and utilizing existing methods and frameworks. Consequently, the Committee completed the formulation of the guidelines by integrating common follow-up approaches found in numerous journals through a systematic review and by considering the domestic healthcare environment.
Level of evidence and feasibility of grading the recommendation
Despite extensive analyses by numerous journals, follow-up strategies for patients who undergo EVT for intracranial aneurysms exhibit substantial variations globally and across different institutions. Regrettably, there is a dearth of guidelines and scientific data defining the optimal approach for the timing and methodology of follow-up in these patients. Consequently, establishing specific recommendations or determining an evidence-based level of guidance is currently not feasible.
Therefore, while unable to provide specific levels of recommendations, the Clinical Practice Guideline Committee of the KoNES has offered recommendations based on an analysis of various papers and considerations of the current situation in the domestic medical field. These recommendations were determined through a deliberative process involving panels of members from multiple societies.
EVIDENCE SUMMARY
Is follow-up imaging necessary after EVT for aneurysms?
Before discussing follow-up imaging after coil embolization for cerebral aneurysms, the necessity for follow-up imaging must be considered. Several large-scale studies have investigated the long-term results of EVT of cerebral aneurysms. In the Cerebral Aneurysm Rerupture After Treatment (CARAT) study, the bleeding rate after coil embolization was 0.11% over a mean follow-up time of 4.4 years. In the International Subarachnoid Aneurysm Trial (ISAT), the annual risk of bleeding after coil treatment of aneurysms was 0.08%. Furthermore, the large-scale single-center BRAT reported no bleeding in the coiling arm after 6 years [11,16,27]. These findings suggest that the risk of rebleeding after coil embolization is relatively low.
However, a crucial aspect of these studies that should not be overlooked is that most patients who underwent coil embolization also underwent regular follow-up examinations. For example, in the ISAT, 88.2% of the patients in the EVT arm (881 patients) underwent follow-up angiography, which was generally performed 6 months after treatment and repeated at varying intervals, as determined by the treating physicians. As a result, 8.3% of the EVT patients in the ISAT received late retreatment without prior rebleeding [4]. Similarly, in the BRAT study, 16.4% of the patients who underwent EVT were retreated. These retreatments were based on findings during the follow-up imaging and were not prompted by rebleeding events [27]. These findings suggest that follow-up imaging plays a crucial role in reducing the rebleeding rate after coil embolization.
A second aspect to consider is that despite being relatively low, the risk of rebleeding is not entirely eliminated. A small percentage of patients experience rebleeding, which can have severe consequences. The final aspect that should be considered is the possibility of de novo aneurysms in patients with intracranial aneurysms, as they can occur in approximately 5–10% of patients. Although these aneurysms may initially be small, some may still pose a considerable risk of bleeding, necessitating treatment [13,14].
Considering factors such as the potential for aneurysm recurrence, the serious consequences of rebleeding, and the occurrence of de novo aneurysms, implementing follow-up imaging after EVT is justified. This approach enables the early detection of lesions and facilitates early intervention to reduce the risk of bleeding.
What would be the optimal timing for the first, intermediate, and long-term follow-ups?
To the best of our knowledge, there are currently no established guidelines or scientific data defining the optimal timing and duration for follow-up imaging after EVT of aneurysms. Different countries and institutions employ different approaches for follow-up protocols. Additionally, when considering the timing of follow-ups, individual patient characteristics, such as the specific characteristics of the aneurysm, patients’ life expectancy, the treatment device, and patients’ psychological factors, must be considered. These factors play substantial roles in the development of appropriate follow-up protocols.
Although establishing universally standardized guidelines remains challenging, various studies have proposed specific timelines for follow-up after EVT. The initial follow-up is recommended within 3–6 months after EVT. This initial follow-up is considered crucial because the first year post-EVT is associated with a higher risk of aneurysm recurrence, with most recurrences occurring during this time [4,14].
The importance of the timing of the first follow-up is widely recognized in the literature, as this allows for surveillance and potential interventions if needed. Although the exact approach for the follow-up after the initial assessment has not been firmly established, some studies indicate that follow-up examinations at 12–24 months and mid-term follow-ups at 3–5 years are commonly included in the follow-up plan [4,14].
However, consistently applying a follow-up imaging plan is difficult, as it can change depending on the specific characteristics of the aneurysm. Patients with risk factors for recurrence, such as ruptured or large aneurysms, wide necks, and incomplete postoperative occlusion, may require more frequent monitoring and extended follow-ups [10,20]. This is unsurprising considering recent data in the literature, which suggest that follow-up periods of 3–5 years may be insufficient to identify significant aneurysm recurrences. These findings raise important questions regarding the optimal monitoring duration for patients after coil embolization.
In a prospective cohort study by Lecler et al. [14], the follow-up observation was conducted for 10 years after the aneurysm coil embolization. They found that sac recanalization occurred in 16 of 129 (12.4%) aneurysms using long-term magnetic resonance angiography (MRA). The researchers also identified Grade 2 classification on the Raymond scale during mid-term MRA (relative risk [RR]: 4.16; 99% confidence interval [CI]: 2.12–8.14) and retreatment within 5 years (RR: 4.67; 99% CI: 1.55–14.03), as risk factors for late recurrence. Additionally, they conducted a systematic review on the same topic, including 15 cohorts comprising 2773 patients with 2902 aneurysms. The review reported bleeding, aneurysm recurrence, and de novo lesion formation rates of 0.7% (99% CI: 0.2–2.7%; I2: 0%; 1/694 patients), 11.4% (99% CI: 7.0–18.0%; I2: 21.6%), and 4.1% (99% CI: 1.7–9.4%; I2: 54.1%), respectively. The review identified initial results of Grade 2 classification on the Raymond scale (RR: 7.08; 99% CI: 1.24–40.37; I2: 82.6%) and aneurysm size >10 mm (RR: 4.37; 99% CI: 1.83–10.44; I2: 0%) as risk factors for late recurrence.
We strongly endorse the conclusion reached by Lecler et al. [14], suggesting that patients in these cases should be considered for longer follow-up periods of ≥10 years.
What imaging techniques should be used for follow-up?
Digital Subtraction Angiography
Digital subtraction angiography (DSA) is considered the most accurate method for assessing the condition of an aneurysm and the parent artery after coiling treatment. It is widely recognized as the gold standard for evaluating aneurysmal occlusion and detecting issues such as intra-aneurysmal flow (aneurysm recurrence) or in-stent restenosis. Therefore, DSA serves as a reference point for comparison with other follow-up imaging modalities.
While DSA remains the most accurate test for detecting recurrent aneurysms, it is an invasive imaging technique that carries various risks, including exposure to ionizing radiation, potential kidney damage from contrast agents containing iodine, the possibility of cerebral thromboembolism, and potential accidental damage to the arteries [7,30,31]. Furthermore, considering that the durability of aneurysm treatments must be confirmed over time through multiple diagnostic studies, the risks associated with DSA follow-up are amplified. In addition, the higher costs associated with DSA and the potential discomfort experienced by patients are relevant factors that need to be considered when designing follow-up regimens.
Magnetic Resonance Angiography
MRA has been increasingly utilized for follow-up evaluations of cerebral aneurysms after EVT because of its noninvasive nature, which helps mitigate some of the risks associated with repeated DSA examinations. Although MRA may have lower sensitivity and be more susceptible to motion artifacts than DSA, there has been a shift toward noninvasive follow-ups using MR imaging (MRI) owing to the inherent risks associated with invasive DSA procedures.
Several noninvasive cross-sectional imaging techniques such as three-dimensional time-of-flight (TOF) MRA and contrast-enhanced (CE) MRA have been developed. These techniques are currently employed in clinical practice for the follow-up evaluation of intracranial aneurysms after coil embolization [22,23]. Meta-analyses conducted by van Amerongen et al. and Menke et al. comparing MRA and DSA as follow-up imaging have reported that MRA shows a moderate-to-high diagnostic performance in comparison to that of DSA [15,29].
However, with advancements in MRI techniques and numerous comparative studies, growing evidence suggests that MRA is comparable to DSA. A recent meta-analysis conducted by Ahmed et al. reported the sensitivity and specificity of MRA in detecting residual aneurysms [1]. The study found that TOF-MRA demonstrated a sensitivity ranging from 86 to 94% and a specificity ranging from 92 to 96%, whereas CE-MRA showed a sensitivity ranging from 92 to 96% and a specificity ranging from 92 to 96%. Furthermore, in their subgroup analysis, Ahmed et al. reported that the sensitivity and specificity of MRA techniques for detecting residual aneurysms in patients undergoing stent-assisted coiling and flow diversion were comparable to those in the coiling only group, which has not been previously reported in the literature. This suggests that MRA can be effective in evaluating residual aneurysms in patients who have undergone these specific treatment modalities [1].
Both CE-MRA and TOF-MRA have been utilized for follow-up evaluation of aneurysms. However, TOF-MRA offers the advantage of not requiring the administration of intravenous contrast agents while maintaining a diagnostic accuracy similar to that of CE-MRA. Therefore, it is recommended as the preferred routine follow-up method to detect any remaining blood flow in patients with intracranial aneurysms treated with endovascular coil occlusion [23,29,31].
The increasing use of intracranial stents has led to concerns regarding the condition of the parent vessel in such cases. Some studies have found that noninvasive techniques such as TOF-MRA and CE-MRA demonstrate high sensitivity in detecting in-stent stenosis. However, both TOF-MRA and CE-MRA show low specificity rates, ranging from 14 to 32% and from 43 to 64%, respectively [2,3].
A recent meta-analysis was performed to determine the potential sources of heterogeneity in the data, such as publication bias, enrollment methods, use of DSA as the reference standard, magnetic field strength in MRI, and study quality. As assessed using the Grades of Recommendation, Assessment, Development, and Evaluation (GRADE) criteria, the analysis demonstrated significantly higher sensitivity and specificity in GRADE 1–2 studies than in GRADE 3–4 studies. Moreover, retrospective studies have shown higher sensitivity and specificity for both TOF-MRA and CE-MRA than those shown by prospective studies [1].
A recent study by Irie et al. introduced a new MRI technique for the follow-up assessment of stent-assisted coiling [12]. They found that silent MRA (GE Healthcare, Milwaukee, Wisconsin, USA) with an ultrashort echo time was effective in visualizing flow within an intracranial stent and assessing the occlusion status of aneurysms. Additionally, another non-enhanced MRA technique called pointwise encoding time reduction with radial acquisition (PETRA) has been introduced, which offers a shorter acquisition time than that of silent MRA and TOF-MRA. These novel techniques show a strong positive correlation with DSA [9,12].
Although these new techniques—silent MRA and PETRA—are not included in existing guidelines, they are widely used in many institutions and have potential as noninvasive follow-up strategies.
Computed Tomography Angiography
Computed Tomography Angiography (CTA) is often used for evaluating intracranial arteries due to its lower cost and shorter scan time compared to MRA. However, it comes with the burden of using contrast agents, unlike MRA. Above all things, image quality can be significantly affected by beam hardening artifacts caused by platinum coil masses. These drawbacks make CTA less suitable for follow-up examinations after coil embolization procedures [26]. It may have limited applications in cases where MRI is not feasible or when assessing vessels located far from the coils. In the near future, the development of algorithms to reduce metallic artifacts may enhance the importance of CTA, addressing some of its current limitations [26].
CONSENSUS ACHIEVEMENT
After the members wrote the completed first draft, which included a proposal for the recommendations, we convened the panel of 40 experts to achieve consensus on the proposed recommendations using the modified Delphi method. Using a 9-point scale modified from the RAND Corporation’s Delphi method, we asked the experts to individually rate each recommendation: a score of 9 indicated strong agreement, whereas a score of 1 indicated strong disagreement. We defined scores of 7–9 as agreement, 4–6 as uncertainty, and 1–3 as disagreement. We defined a consensus on a recommendation as agreement by ≥75% of the experts. Additional Delphi rounds were conducted for recommendations with an agreement rate of <75%.
During the initial Delphi round, a consensus was reached on seven recommendations, and the overall scores and agreement rates were consistently high, exceeding 90%. However, specific items related to invasive methods, such as, in the case of stent-assisted coiling, “DSA is considered the gold standard for follow-up evaluation,” and “it is recommended that DSA be performed as a mandatory procedure during the first follow-up after stent-assisted coiling, followed by comparative analysis with MRA,” showed lower agreement rates of 63% and 68%, respectively. Consequently, the seven recommendations were revised and consolidated into five recommendations for the second Delphi round, wherein they were re-evaluated. Through this iterative process, an agreement rate of >90% was achieved. The final draft of the guidelines was thoroughly reviewed and approved by the participating academic societies.
RECOMMENDATIONS
Fig. 1 summarizes the consensus on follow-up imaging after EVT of intracranial aneurysms. The consensus details are as follows:
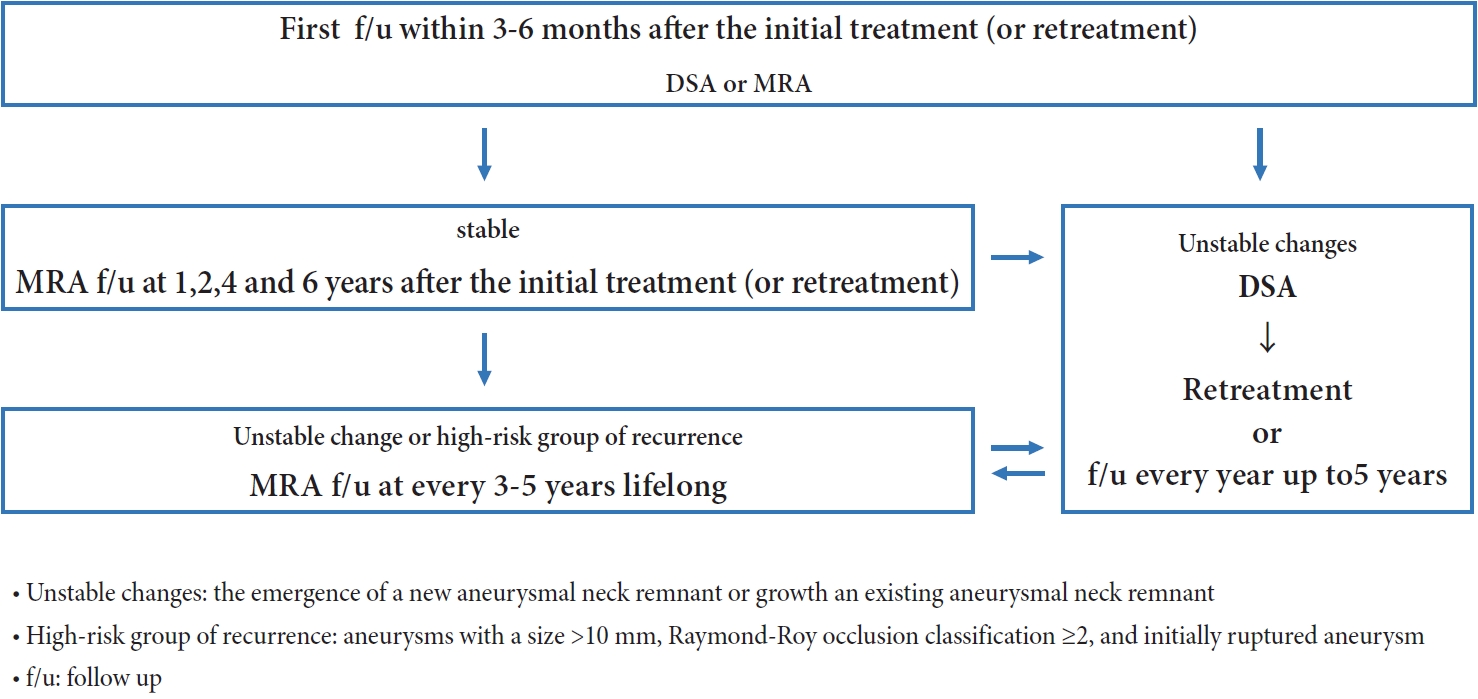
Summary of the expert panel consensus for follow-up imaging after endovascular treatment (EVT) of intracranial aneurysms
1. For patients who have undergone EVT (simple coil embolization or stent-assisted coil embolization) for intracranial aneurysms, it is recommended to schedule the initial follow-up imaging within 3–6 months.
2. DSA remains the gold standard, but noninvasive imaging modalities such as CE-MRA or TOF-MRA can be considered as alternatives during the first imaging follow-up.
3. Following the initial follow-up imaging, it is recommended to consider mid-term imaging follow-ups at 1, 2, 4, and 6 years after the initial treatment.
4. If noninvasive imaging reveals unstable changes in the treated aneurysms, such as the emergence of a new aneurysmal neck remnant or the growth of an existing aneurysmal neck remnant, it is recommended to consider DSA for further evaluation and to discuss the potential need for retreatment.
4-1 For patients with identified unstable changes who have opted for observation rather than for retreatment, it is advisable to undergo more frequent noninvasive imaging (such as CE-MRA or TOF-MRA) examinations, compared with that for individuals without identified unstable changes. This includes annual imaging follow-ups for up to 5 years.
4-2 Patients who have undergone retreatment owing to unstable changes should be treated as if they were receiving initial treatment and should follow the same follow-up imaging schedule. (It is recommended to schedule the initial follow-up imaging within 3–6 months).
5. For patients who exhibit unstable changes or are at high risk of recurrence, it is recommended to consider late-term follow-up imaging every 3–5 years for lifelong monitoring. High risk for recurrence typically includes aneurysms with a size >10 mm, aneurysms with a Raymond-Roy occlusion classification of Grade ≥2, and initially ruptured aneurysms.
LIMITATIONS AND FUTURE DIRECTIONS
Although the developed guidelines provide valuable recommendations for the follow-up of patients with intracranial aneurysms treated using endovascular methods, the limitations and future directions of these guidelines must be acknowledged.
First, the current guidelines are based on available evidence and a consensus reached by an expert panel. However, they only include information on EVT of intracranial aneurysms using coils and do not cover follow-up strategies after the implementation of actively performed procedures such as flow diverters and flow disruptors. With advances in medical knowledge and technology, new evidence may emerge, necessitating periodic updates to ensure that the guidelines remain relevant and reflect the latest developments in the field.
Second, the guidelines primarily focus on imaging modalities such as CE-MRA and TOF-MRA. Although these methods are commonly used and have shown promising results, there may be emerging imaging technologies or techniques that could enhance the accuracy and effectiveness of follow-up assessments. Future research should explore the utility of these novel approaches and consider their potential inclusion in the guidelines.
Third, the current guidelines primarily address a follow-up period of up to 6 years after the initial treatment. However, long-term outcomes and optimal follow-up strategies for patients beyond this timeframe remain poorly defined. Further studies are warranted to evaluate the long-term efficacy and safety of different follow-up intervals and imaging modalities for the lifelong monitoring of patients with intracranial aneurysms.
Finally, the guidelines should consider the individual characteristics and preferences of patients. Tailoring the follow-up approach according to patient-specific factors such as age, comorbidities, and treatment outcomes could help optimize the monitoring process and improve patient outcomes.
In conclusion, although the current guidelines provide valuable recommendations, acknowledging their limitations and actively pursuing future research and updates will ensure that they serve as valuable resources for clinicians managing patients with intracranial aneurysms.
Supplementary figure and table
Consensus based on the modified Delphi method
Notes
Disclosure
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
