Undeflatable balloon guide catheter (BGC) during endovascular procedure: Rescue strategy
Article information
Abstract
The use of a balloon guide catheter (BGC) in the endovascular management of acute ischemic stroke is known to improve the efficacy and efficiency of the procedure by reducing the risk of distal embolization. During the procedure, the balloon of the catheter causes a temporary arrest of cerebral blood flow. However, failure of the balloon to deflate during the BGC procedure can result in catastrophic complications, including aggravated hypoxic damage.
This paper aims to share the resolution and methodological analysis of our experience with BGC balloon deflation failure, which was confirmed by a reproducible experiment under similar conditions.
INTRODUCTION
Following the recent publication of five randomized trials (MR CLEAN, SWIFT PRIME, EXTEND-IA, ESCAPE, REVASCAT, THRACE) [8,20], mechanical thrombectomy, along with medical management, was established as the standard treatment for acute ischemic stroke caused by large vessel occlusion. Delays in performing mechanical thrombectomy for the purposes of awaiting results of intravenous alteplase for treatment of acute cerebral infarction are discouraged in the 2019 AHA/ASA guideline [8], as are recommendations to perform immediate mechanical thrombectomy on patients who visit the hospital within 6 h of the onset of symptoms and do not meet the exclusion criteria [8,20]. If the eligibility criteria for DAWN (within 6–16 h) or DEFUSE 3 (within 16–24 h) trials are met [1,17,20], mechanical thrombectomy is possible even in cases in which more than 6 h have passed since the onset of symptoms or the last normal time is unknown.
In the treatment of acute ischemic stroke, various mechanical thrombectomy strategies, such as stent retrieval technique and direct aspiration thrombectomy [20,27], are used in conjunction with novel devices such as balloon guide catheters (BGC) or large-bore distal-access catheters [3,10,16,31] for increased efficacy and efficiency [20]. The BGC, in particular, is known to lower the risk of distal embolization during the procedure and improve flow reversal effects by the deployment of a balloon, which causes the arrest of cerebral blood flow [7,21,28], Such an advantage has been reported in two recent clinical studies to increase recanalization rate and improve neurological outcome [6,16,27,28].
However, failure of the balloon to deflate during the BGC procedure can result in catastrophic complications, including aggravated hypoxic damage. This paper aims to share the resolution and methodological analysis of our experience with BGC balloon deflation failure, which was confirmed by a reproducible experiment under similar conditions.
CASE PRESENTATION
A 104-year-old female patient presented to the emergency department with a chief complaint of altered consciousness and right hemiparesis. She was apparently normal 5 h prior to the presentation. No cerebral hemorrhage was detected on non-contrast brain computed tomography (CT). The patient had an Alberta Stroke Program Early CT Score (ASPECTS) of seven and a National Institutes of Health Stroke Scale (NIHSS) score of 15. An occlusion in the left proximal M1 segment was detected (Fig. 1A) on CT angiography of the brain, and mechanical thrombectomy was performed immediately (Fig. 2A).
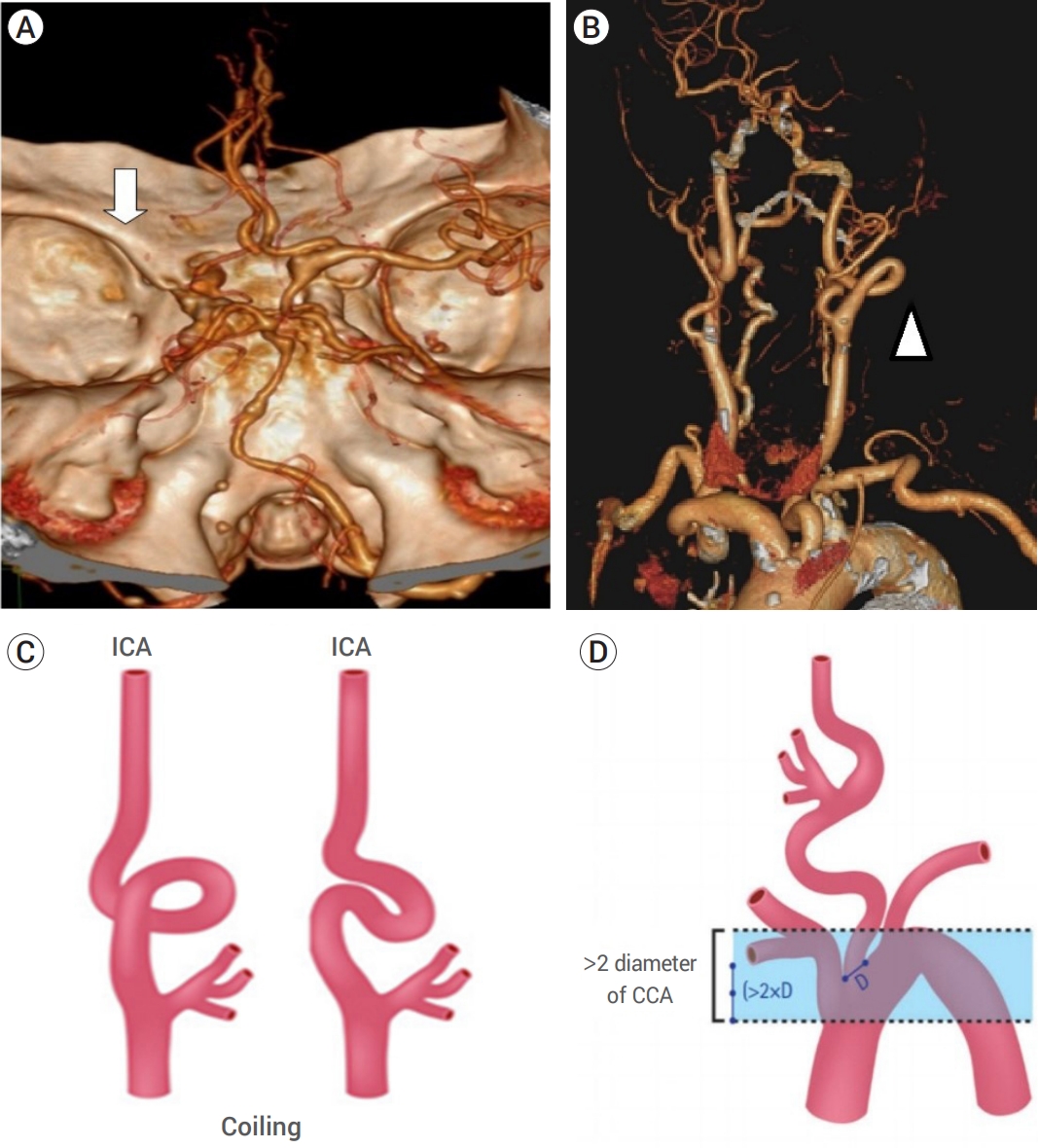
The anatomic features are known to be unfavorable for endovascular procedures. (A) Shows computed tomographic angiography of the brain (white arrow) shows flow arrest caused by left middle cerebral artery occlusion. (B) Shows the aortic arch and the curvature of the internal carotid artery (ICA) (head of the white arrow) in the patient studied. (C) Shows the “coiling” feature of proximal ICA with extreme tortuosity, and (D) shows the vertical distance between the innominate artery and the top of the aortic arch, which was greater than twice the diameter of the left common carotid artery [2].

Cerebral angiography in the patient studied. (A) Shows the anteroposterior view of cerebral angiography after stent deployment for stent retrieval. Because of the recanalization of the left middle cerebral artery, distal flow is visible. However, re-occlusion occurred during stent retrieval. (B) Shows the lateral view of the balloon guide catheter (BGC) in the proximal part of the coiling segment in the left ICA. The sudden deflation of the BGC failed, and there was prolonged arrest of intracerebral flow. (C) Successful deflation was achieved, and intracerebral flow restored following a series of processes involving the excision of a proximal portion of the BGC and the passage of cutting balloon through the BGC.
Procedure for mechanical thrombolysis
The procedure was performed under local anesthesia. Flowgate BGC (8-Fr, Stryker Neurovascular, Fremont, CA, USA) (Fig. 3A) was inserted through the femoral sheath and placed proximally near the left internal carotid artery (ICA). During the procedure the balloon of BGC was inflated with 1:1 mixed contrast solution. Left ICA angiography revealed occlusion of the left proximal M1 segment, and stent retrieval was performed using the Trevo XP ProVue stent (4 mm×20 mm, Stryker Neurovascular, Fremont, CA, USA). Although the left middle cerebral artery (MCA) was recanalized at the time the stent was deployed, several re-occlusions occurred following stent retrieval. Additional attempts were made to recanalize the MCA using the ADAPT [12,23] and Solumbra techniques [29,30] with the AXS Catalyst 6 Distal Access Catheter (Stryker Neurovascular, Fremont, CA, USA), but the outcomes remained the same. During the procedure, the balloon of the BGC failed to deflate (Fig. 2B). Although aspiration with a syringe was attempted several times to de-balloon the BGC, it failed to de-balloon and intracerebral blood flow arrest persisted with the balloon fixed to the proximal ICA.
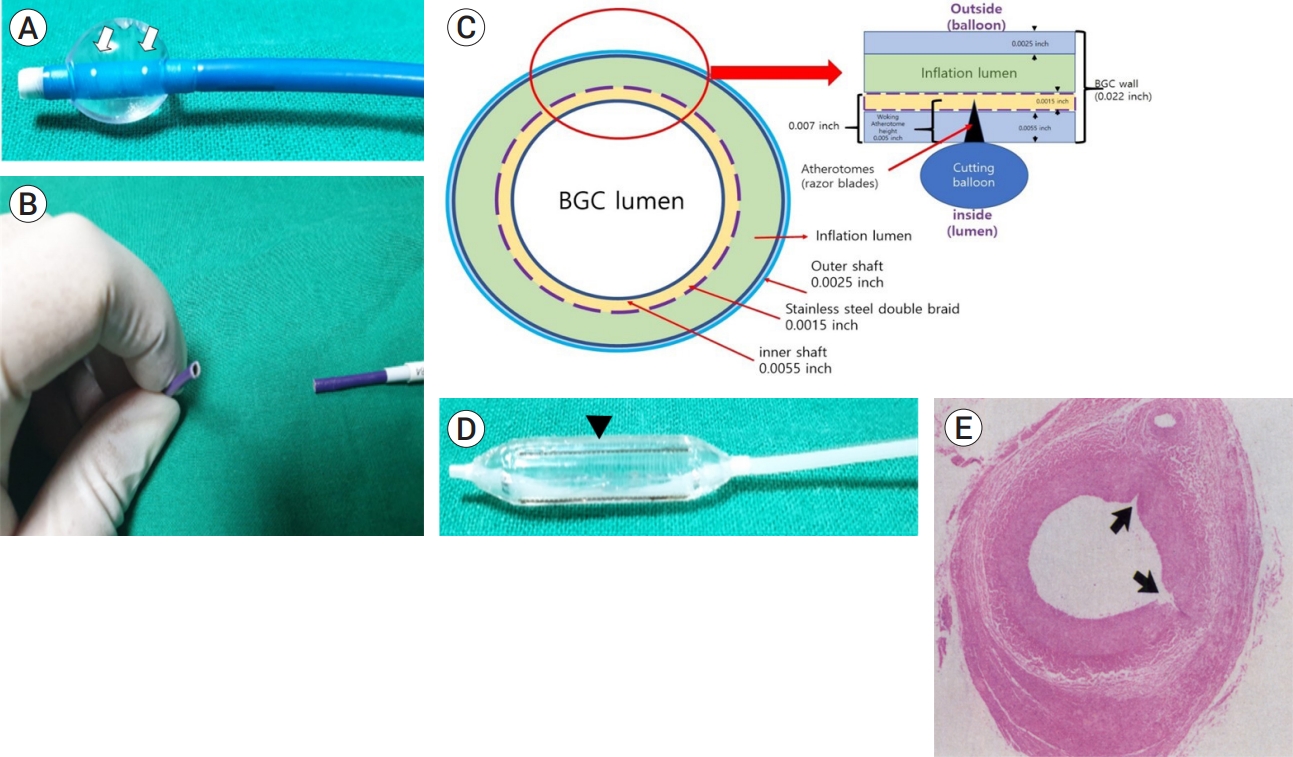
The structural feature of the balloon guide catheter (BGC) and cutting balloon. (A) Is an inflated BGC; it shows leakage of the contrast media from the two white dots (white arrows) above the region in which the balloon is inflated. (B) Shows the excised surface of the BGC, which is relatively clean, and the structure has not been crushed or deformed. (C) Shows the cross-sectional structure of the excised BGC. The balloon of the BGC is located outside the 0.022-inch catheter wall, which consists of an outer shaft, stainless metal braid and inner shaft with thickness of 0.0025-, 0.0015- and 0.0055-inches, respectively. Because, the thickness of the catheter from the lumen to the inflation lumen is 0.007 inches, the 0.005-inch blade of the cutting balloon does not reach the balloon of the BGC, or the inflation lumen. (D) Shows three to four razor blades (at the heads of the black arrows) present on the surface of the cutting balloon. (E) Shows a cross-section of the transverse microincision made by the cutting balloon on the vessel lumen (black arrow) [4]. The BGC wall was expected to be damaged by the blades of the cutting balloon, resulting in the deflation of the balloon.
Troubleshooting
To enable immediate de-ballooning, a transverse incision was made along the proximal part of the BGC (Fig. 3B). Because there is a channel embedded within the wall of the BGC that allows the balloon to inflate (Fig. 3C), it was assumed that removing the catheter would expose the inflation channel or space, releasing the contrast solution in the balloon and ultimately de-ballooning it. However, deflation once again failed, despite the transverse excision of the proximal portion of the BGC. An intraluminal approach by passing a cutting balloon through the BGC was attempted to deflate the balloon. A cutting balloon is a device with three to four microsurgical blades attached to the balloon surface (Fig. 3D) [5,25] that is designed to create microincisions in surrounding structures when inflated (Fig. 3E).
A 5-mm peripheral cutting balloon (5.0 mm×2.0 cm, Boston Scientific Corporation, Marlborough, MA, USA) with a diameter slightly smaller than the proximal ICA was used to introduce the device into the BGC. The cutting balloon was inflated after being inserted and placed near the BGC balloon, and the process was monitored in real-time using fluoroscopy. While the cutting balloon was being inflated, the balloon of the BGC remained inflated. However, a few seconds after the cutting balloon was inflated, the balloon of the BGC gradually decreased in size and eventually became completely deflated. As a result, the BGC was successfully removed from the ICA and the left ICA flow was recovered from a state of flow arrest (Fig. 2C). All it takes 15 minutes to bail out and there was no interval change in the patient’s neurologic condition.
DISCUSSION
Endovascular treatment has recently been established as an essential treatment for acute ischemic stroke [8,20]. BGC, in particular, has been widely used to improve the success rates of recanalization and shorten procedure time during stent retrieval thrombectomy [16,28].
Although there are various complications associated with endovascular procedures, there have been reports of coronary artery intervention for complications related to deflation failure [13,24,26]. The EuroPCR group has proposed a number of rescue therapeutic options in cases of non-deflatable coronary balloons (www.pcronline.com) [19].
Brinjikji et al. reported groin complications (groin or retroperitoneal hematoma, femoral artery bleeding, etc.) caused by large groin sheaths [6], whereas catheter instability, carotid artery dissection, and difficult aortic arch have been identified as potential complications [9]. However, there have been no reports of balloon deflation failures or discussions regarding potential solutions in the case of balloon deflation failure as observed in this case.
Balloon deflation failure is a very rare complication of BGC that occurred in this case, and the potential causes and solutions were analyzed. First, the composition ratio of the contrast solution used in the balloon may influence deflation failure. A 1:1 dilution of the contrast has been suggested as an optimal concentration considering visibility and balloon deflation time [11,14]. The contrast solution used in this case was also 1:1. Second, potential mechanical factors may also influence the deflation of the balloon. However, the possibility of mechanical error was excluded in this case, as the deflation failure occurred after several trials of successful ballooning and de-ballooning. The anatomical factors of the patient may also influence the deflation of the BGC. The patient in this case was an adult 104-year-old adult with a very steep vasculature entry angle and tortuous morphology (Fig. 1B). Her left ICA had a coiling morphology with a circular configuration, and her aorta had a vertical distance between the innominate artery and the top of the aortic arch that was more than twice the diameter of the left common carotid artery, which is considered unfavorable for an endovascular approach procedure (Fig. 1C, D) [2,15]. Even if the BGC was safely positioned at the proximal ICA before the circular segment, the position of the BGC may have shifted incidentally during the repetitive ballooning and de-ballooning process and stent retrieval. As a result, structural changes or damage to the inflation lumen of the BGC, such as torsion or kinking, could have occurred, leading to deflation failure.
Given the mechanism by which the balloon deflated after several attempts, it was surprising why the excision of the proximal portion of FlowGateTM did not cause the balloon to deflate. The FlowGateTM structure includes a lumen-shaped channel (inflation lumen) that is used to inflate the BGC balloon through an external injection of contrast solution (Fig. 3C). Because this lumen is embedded within the catheter wall, excision of the catheter should result in immediate drainage of the contrast solution and subsequent deflation of the balloon, though this did not happen in this study. Furthermore, given that deflation occurred immediately after the introduction and inflation of the cutting balloon, it is unclear whether the deflation was caused by the cutting balloon based on the profiles of the BGC and cutting balloon.
The cutting balloon was first invented by Dr. Peter Barath and was mainly used for the treatment of coronary artery diseases [4]. Unlike conventional balloon catheters, cutting balloons have three to four 0.005-inch atherotomes (razor blades) on the balloon surface (Fig. 3D) [4,5]. When the cutting balloon is inflated, the blades are designed to disrupt the fibroelastic continuity of the neointimal hyperplasia by making sharp transverse microincisions (Fig. 3E) [4,18,22]. Although deflation was attempted by inserting the cutting balloon into the BGC, it was impossible to deflate the balloon in the BGC with the 0.005-inch blade, due to the structure of the BGC wall, which requires a blade of at least 0.007 inches (Fig. 3C).
To simulate the situation, a FlowGateTM was inserted into a rubber tube with the thickness of a blood vessel and ballooned with a 1:1 contrast solution. The cutting balloon was then inflated and monitored in real-time using fluoroscopy, but the balloon in the BGC did not deflate. The BGC balloon did not rupture despite several attempts with different positions of the cutting balloon and the application of sufficient pressure to cause the cutting balloon to rupture the BGC balloon (Fig. 4A, B).

The methodological analysis of our experience with balloon guide catheter (BGC) balloon deflation failure, which was confirmed by a reproducible experiment under similar conditions. (A) Shows the introduction of the cutting balloon in the inflated BGC. (B) No deflation was observed in the BGC balloon, although sufficient pressure was applied for the cutting balloon to rupture the BGC. The contrast media is leaking due to a rupture in the cutting balloon (white arrow). (C) Shows the BGC inflated inside a rubber tube. The balloon did not deflate despite the excision of the proximal portion of the BGC. This may be attributed to the luminal collapse caused by the flexure in the catheter. (D) After excising the proximal portion of the BGC and straightening the kink in the flexure of the catheter, luminal patency is obtained, and deflation occurs automatically within 30 s.
The proximal portion of the BGC was then excised. Once a straight BGC was prepared and inflated in the aforementioned rubber tube, followed by excision of the proximal portion of the catheter, solution leakage occurred in the excised portion of the BGC, resulting in complete deflation within 30 s. The time was then recorded, while the BGC was used to create flexures ranging from 0 to 90°. It was discovered that at a 90° flexure, the balloon had only partially deflated even after 5 min, in contrast to the 30 s it took for the balloon to deflate at a 0° flexure (Fig. 4C, D).
In conclusion, the findings show that the cutting balloon does not cause damage to the BGC wall and that deflation is possible by excising a proximal portion of the BGC. However, it was demonstrated that in the presence of flexure in the BGC, the deflation time may be delayed, or only partial deflation may occur. Kinks in the catheter were found to be present in cases like this, where deflation failed to occur even after the proximal portion of the BGC was excised. As a result, inserting a stiff device into the interior of the BGC to straighten the collapsed catheter lumen caused by the kinking may be required as an additional procedure.
CONCLUSIONS
The BGC balloon deflation failure observed in this case is an extremely rare and unknown complication in the neurovascular field that can lead to fatal and catastrophic outcomes if not addressed immediately. Although rare, if the balloon fails to deflate during a BGC procedure, knowing how to restore luminal patency with a stiffer device and excising the BGC will help to successfully deal with this rare complication.
Notes
Disclosure
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
