 |
 |
| J Cerebrovasc Endovasc Neurosurg > Epub ahead of print |
Abstract
Objective
Stroke is the second-leading cause of death globally. Intracranial atherosclerotic stenosis (ICAS) represents 10-15% of ischemic strokes in Western countries and up to 47% in Asian countries. Patients with ICAS have an especially high risk of stroke recurrence. The aim of this meta-analysis is to reassess recurrent stroke, transient ischemic attack (TIA), and other outcomes with stenting versus best medical management for symptomatic ICAS.
Methods
The search protocol was developed a priori according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The OVID Medline, Embase, Web of Science, and Cochrane Library databases were searched from inception to August 14th, 2022.
Results
This Meta-analysis Included Four Randomized Controlled Trials (Rcts), With A Total Number Of 991 Patients. The Mean Age Of Participants Was 57 Years. The Total Number Of Intracranial Stenting Patients Was 495, And The Number Of Medical Treatment Patients Was 496. The Included Studies Were Published Between 2011 And 2022. Two Studies Were Conducted In The Usa, And The Other Two In China. All Included Studies Compared Intracranial Stenting To Medical Treatment For Icas.
Conclusions
In patients with ischemic stroke due to symptomatic severe intracranial atherosclerosis, the rate of 30-day ischemic stroke, 30-day intracerebral hemorrhage, one-year stroke in territory or mortality favored the medical treatment alone without intracranial stenting. The risk of same-territory stroke at last follow-up, disabling stroke at last follow-up, and mortality did not significantly favor either group. Intracranial stenting for atherosclerosis did not result in significant benefit over medical treatment.
Stroke is the second-leading cause of death globally and the leading cause of death in China [11,27]. Intracranial atherosclerotic stenosis represented 10-15% of ischemic strokes in Western countries [21], and up to 47% in Asian countries in 2009 [20]. Patients with intracranial atherosclerotic stenosis have an especially high risk for stroke recurrence [3,12], which has long prompted the study of transluminal percutaneous angioplasty and/ or stenting [4,9,13,25]. However, the Stenting vs. Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis (SAMMPRIS) trial was discontinued early because of a significantly higher rate of stroke or death with stents compared to medical therapy (14.7% versus 5.8%; P=0.002) [5]. Similarly, the Vitesse Intracranial Stent Study for Ischemic Stroke Therapy (VISSIT) [24] and a randomized single-center trial in China14) showed no advantage of stenting compared to medical treatment.
Subsequently, some prospective multi-center registries suggested better patient selection (i.e., excluding patients with ischemic events due to perforator blockage and requiring a longer period from the ischemic event). Moreover, experienced interventionalists and comprehensive teams have been suggested to decrease the periprocedural risk of stenting from 14.7% to between 2.0% and 4.3% [2,10,14]. A prospective registry of 100 stented patients with refined criteria reported a stroke at 30 days or a death rate of 2.0% [10]. The Wingspan Stent System Post Market Surveillance Study (WEAVE) suggested a similarly low rate of periprocedural complication (2.6%) [2]. These lower risks of periprocedural complications in the modern era have prompted revisitation of randomized controlled trial (RCT) methodology.
The China Angioplasty and Stenting for Symptomatic Intracranial Severe Stenosis (CASSISS) was a multicenter, randomized, open-label study. It assessed the impact of stenting versus medical therapy on death and stroke in patients with a transient ischemic attack (TIA) or non-disabling ischemic stroke with severe intracranial atherosclerotic stenosis admitted to high-volume medical centers, using careful selection of patients. The aim of this meta-analysis is to reassess recurrent stroke, TIA, and other outcomes with stenting versus best medical management for symptomatic intracranial stenosis including these new data in the modern era.
The search protocol, including research questions and inclusion and exclusion criteria, was developed a priori according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The OVID Medline, Embase, Web of Science, and Cochrane Library databases were searched from inception to August 14th, 2022. References of included publications were searched manually for other relevant papers. To identify studies of stenting versus best medical management for symptomatic intracranial stenosis, the following key words were used in combination: “ICAD”, “intracranial stenosis”, “intracerebral stenosis”, “middle cerebral artery stenosis”, “MCA stenosis”, “intracranial stenting”, “intracerebral stenting”. The search was limited to articles in humans, in patients ≧18 years of age, and in English.
Studies were included if they utilized a randomized design reporting treatment-stratified clinical outcomes in symptomatic intracranial stenosis, comparing stenting versus best medical management. Studies which did not compare stenting with best medical management were not included. Studies which were not randomized control trials, such as prospective or retrospective studies comparing the two treatments, were not included. Single arm studies were not included. Studies which included patients with asymptomatic intracranial stenosis were excluded. If there were multiple studies from overlapping cohorts of patients, such as those from the same center, only studies with the largest cohort of patients were included. Exclusion criteria included case reports, case series with less than five patients, review articles, conference abstracts, animal studies, and non-peer reviewed publications, all other applicable articles were included Fig. 1.
Two reviewers (J.C.K., A.Y.A.) performed screening and any discrepancies were independently verified by another reviewer (A.A.D.). The primary outcomes were stroke rates following treatment, specifically, 30-day stroke or death, one year stroke in territory or death, and last follow-up stroke in territory, as defined by the authors of the original study. Other outcomes included the 30-day hemorrhage rate, disabling stroke at last follow-up, and death at last follow-up. The following data was extracted from each study: study design, country of origin, patient eligibility, inclusion/exclusion criteria, age and sex of patients, and the outcome measures outlined above. The quality of all eligible studies was evaluated independently and in duplicate by two reviewers (J.C.K., A.Y.A.).
All data were analyzed using R Version 4.2.1 to compute the risk ratio (RR) for the included studies; all included studies were randomized control trials (RCT). No different conditions were found in the same outcomes. Heterogeneity was assessed with Q-statistics and was considered significant when I2 >50% or P-value <0.05. The corresponding 95% confidence intervals (CI) of pooled effect size were calculated using a fixed-effects model (common-effect model), fixed-effects model, and common-effects model are the same; however, in the new update of metabin and forest libraries in R Version 4.2.1, the fixed term was replaced with common. The common-effects model was selected since the heterogeneity in our analysis was not statistically significant. Egger’s regression was not assessed since the number of included studies is less than ten. The included studies assessed six different outcomes: 30-day stroke or death, 30-day intracerebral hemorrhage, one-year stroke in territory or death, stroke in a territory at the last follow-up (one-year and three-year), disabling stroke at the last follow-up, and death at the last follow-up.
This meta-analysis included four RCT studies, with a total number of 991 patients. The mean age of participants is 57 years old (SD=23). The total number of intracranial stenting patients was 495 participants, and the number of medical treatment patients was 496. The included studies were published between 2011 and 2022. Two studies were conducted in the U.S, and the other two in China. All the included studies compared intracranial stenting to medical treatment for intracranial atherosclerosis. The baseline characteristics of the included studies are listed in Table 1. Results of the included studies extraction are listed in Table 2.
Based on the common-effect model meta-analysis, the rate of stroke or death within 30 days significantly favored the medical treatment group over the stenting group (RR: 2.37, 95% CI [1.47; 3.81]), and the heterogeneity was not statistically significant (I2=0%, p=0.95), Fig. 2.
The rate of 30-day intracerebral hemorrhage (ICH) significantly favored the medical treatment group over the stenting group (RR: 13.44, 95% CI [2.58; 69.88]), and the heterogeneity was not statistically significant (I2=0%, p=0.90), Fig. 3.
The rate of one-year stroke in territory or death significantly favored the medical treatment group over the stenting group, and the stenting group had a 61% higher relative risk for stroke in territory or death compared to the medical group (RR: 1.61, 95% CI [1.18; 2.20]); the heterogeneity was not statistically significant (I2=0%, p=0.50), Fig. 4.
The rate of stroke in territory at the last up follow-up did not significantly favor either the stenting group or medical treatment group after one-year follow-up or after three-year follow-up (RR: 1.70, 95% CI [0.98; 2.98]), (RR: 1.37, 95% CI [0.99; 1.89]). However, the overall estimate combination for one-year and threeyear effects significantly favored the medical treatment group over the stenting group. The stenting group had a 44% higher risk of stroke in territory compared to the medical group (RR: 1.44, 95% CI [1.09; 1.91]). The heterogeneity was not statistically significant (I2=0%, p=0.44), Fig. 5. This statistical significance after combination supports that the event is mainly intervention dependent rather than time dependent.
The rate of disabling stroke at the last follow-up did not significantly favor either the stenting group or medical treatment group (RR: 1.30, 95% CI [0.86; 1.95]), and the heterogeneity was not statistically significant (I2=0%, p=0.91), Fig. 6.
The mortality rate at the last follow-up did not significantly favor either the stenting group or medical treatment group (RR: 1.41, 95% CI [0.77; 2.59]), and the heterogeneity was not statistically significant (I2=10%, p=0.33), Fig. 7.
The CASSISS trial suggested that stenting plus medical therapy, compared with medical therapy alone, did not lead to a significant difference in the risk of stroke or subsequent death within 30-day or stroke in territory beyond 30-day to one year. The results of all pre-determined secondary outcomes also revealed no significant differences.
Although efforts have been made to reduce perioperative complication rates by credentialling operators and sites as well as refining patient selection, these updated results demonstrated no clinical benefit from stenting over medical therapy for treating patients with symptomatic severe intracranial atherosclerotic stenosis. Results from this study, as well as previous trials [5,14,24], support the recent American Academy of Neurology Practice Advisory on preventing stroke for symptomatic intracranial atherosclerosis of major arteries [17] that recommends aggressive medical therapy instead of stenting for patients with symptomatic severe intracranial atherosclerotic stenosis.
Compared to earlier randomized trials [5,14,24], the one-year cumulative stroke risk or mortality risk in both the stenting and medical therapy groups was significantly lower. The main reason for the difference is probably the exclusion of patients with ischemic symptoms within three weeks of enrolling in the study. These patients were probably at the highest risk for 30-day stroke or death. All prior studies included patients who were not required to respect the interval between disease onset and registration. Other reasons may be differences in age and ethnicity. The mean age was 56 years in this study compared to 60 years in SAMMPRIS and 62 years in VISSIT. SAMMPRIS and VISSIT were predominantly black and white, and the current study population was predominantly Chinese.
The rate of 30-day events after stenting was higher in the SAMMPRIS and VISSIT studies, resulting in an early termination. The current study’s 30-day event rate was also numerically higher among the stenting group. While much of the reduced procedural risk seen in this study may be attributable to patient selection, other factors are probably involved.
Firstly, CASSISS chose high-volume clinical sites and used an introductory phase for accredited interventionalists to ensure their experience with stent application. The association of a lower risk of complication with higher volume centers has also been demonstrated in SAMMPRIS [26] and the WEAVE registry [2]. Also, the National Institutes of Health registry [15] suggests how important experience is in performing intracranial stent procedures [22]. Differences in peri-procedural care may also be a key differentiator for high-volume centers. Secondly, careful patient selection is felt to diminish the periprocedural risk. All patients in this study had an MRI or CT scan at screening, and those with a perforator stroke alone without artery-to-artery embolism or distal hypoperfusion were excluded. This exclusion criterion may have decreased the risk of perforation associated with the stenting procedure. The SAMMPRIS trial included 23% of patients with perforator stroke [18].
A post hoc analysis of SAMMPRIS data revealed that the majority of periprocedural strokes in the stenting group were perforator strokes (71.42%) [1,8]. Thirdly, the timing was also linked to safety results for the stent [26]. Recent studies have shown that stenting within three weeks may present a greater procedural risk [19,23]. This study involved patients whose delay between the index event and stenting was greater than three weeks (median time, 35 days), and this was much longer than that of SAMMPRIS (median time, 7 days) [5] and VISSIT (median time, 9 days) [24]. Increased risk of complications of early stenting could be related to plaque detachment or reperfusion injury [5,7,15].
One of the factors that possibly explains the lack of superiority from stenting over medical therapy is the not insignificant periprocedural risk of complications. The symptomatic 30-day ICH rate was numerically higher in the stenting group compared to the treatment group (2.3% [4/176] vs 0% [0/182]). In the stenting group, the risk of ICH may be related to vessel perforation by the microwire during the stenting process. Apart from device limitations, the endovascular approach for treating intracranial stenosis can pose technical challenges. Furthermore, patient’s with ICAD often have tortuous vasculature, which can be difficult to navigate. Crossing narrow diseased vessels can also disrupt atherosclerotic plaques. Furthermore, periprocedural management can have associated risks, such as maintaining an elevated goal systolic blood pressure to prevent hyperperfusion after a procedure [2,10]. This may be an important topic of future study since there were two reperfusion hemorrhages after intervention in the stenting group. Another factor contributing to the lack of benefit from stenting may be the lower risk of ischemic stroke in the medical therapy group as medical management approaches have improved over time.
During the three-year follow-up, there were no significant differences between the two groups. These data suggest that although the periprocedural risk in the stenting group could be reduced to as low as the 30-day level in the medical therapy group alone, stents may not offer long-term advantages over medical therapy. As noted above, the low rate of events in the medical therapy group is likely associated with a longer time interval after the onset of the last symptom at randomization (median, 35 days in the present study compared to 7 days in SAMMPRIS [5] and 15 days in VISSIT [24]).
In three studies, most strokes occurred in a relatively short period after the index event, and no other ischemic events occurred in the second or third year [5,6,16]. In the MyRIAD (Mechanisms of Early Recurrence in Intracranial Atherosclerotic Disease) study, which included patients presenting with symptoms of intracranial atherosclerosis resulting in 50%-99% stenosis, five of the nine ischemic strokes in the same territory (55.6%) occurred before the first follow-up visit of 6-8 weeks [16]. This trend was also apparent in previous randomized trials [5,6,16].
In patients with ischemic stroke due to symptomatic severe intracranial atherosclerosis, the rate of 30-day stroke, 30-day intracerebral hemorrhage, and one-year stroke in territory or mortality was favorable in the medical treatment without intracranial stenting group. The risk of stroke in territory at last follow-up, disabling stroke at last follow-up, and mortality was not significantly different between groups in this updated meta-analysis, and further honing of patient selection may be needed to revisit stenting. Limitations of this meta-analysis are due to the smaller number of included studies, and further clinical studies are needed to provide stronger evidence for intracranial stenting in atherosclerotic patients.
Fig. 1.
PRISMA flowchart. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses
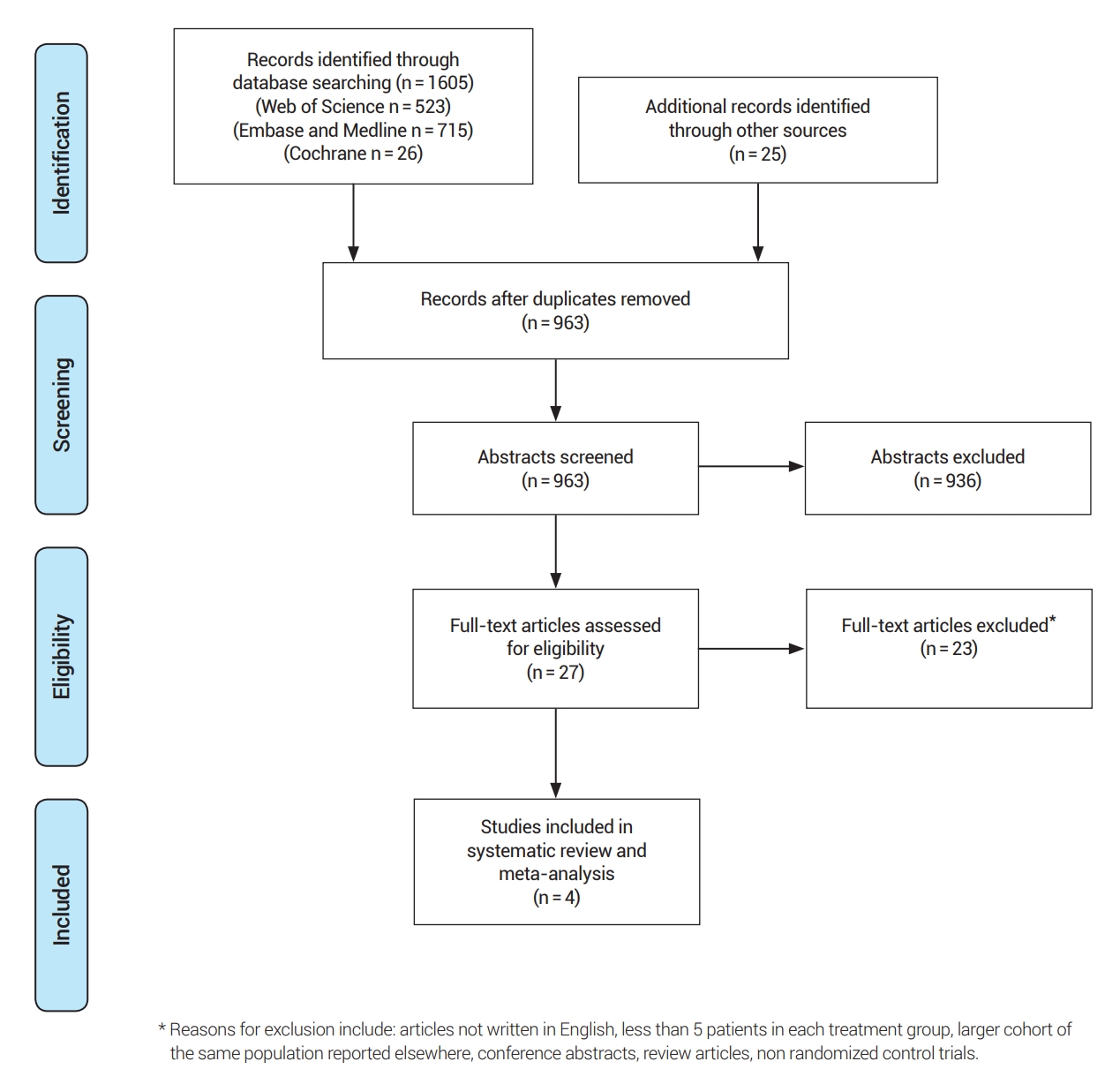
Fig. 2.
Forest plot for 30-day stroke or death. RR, risk ratio; CI, confidence intervals; CASSISS, China Angioplasty and Stenting for Symptomatic Intracranial Severe Stenosis; VISSIT, Vitesse Intracranial Stent Study for Ischemic Stroke Therapy; SAMMPRIS, Stenting vs. Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis

Fig. 3.
Forest plot for 30-day intracerebral hemorrhage. ICH, intracerebral hemorrhage; RR, risk ratio; CI, confidence intervals; CASSISS, China Angioplasty and Stenting for Symptomatic Intracranial Severe Stenosis; VISSIT, Vitesse Intracranial Stent Study for Ischemic Stroke Therapy; SAMMPRIS, Stenting vs. Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis
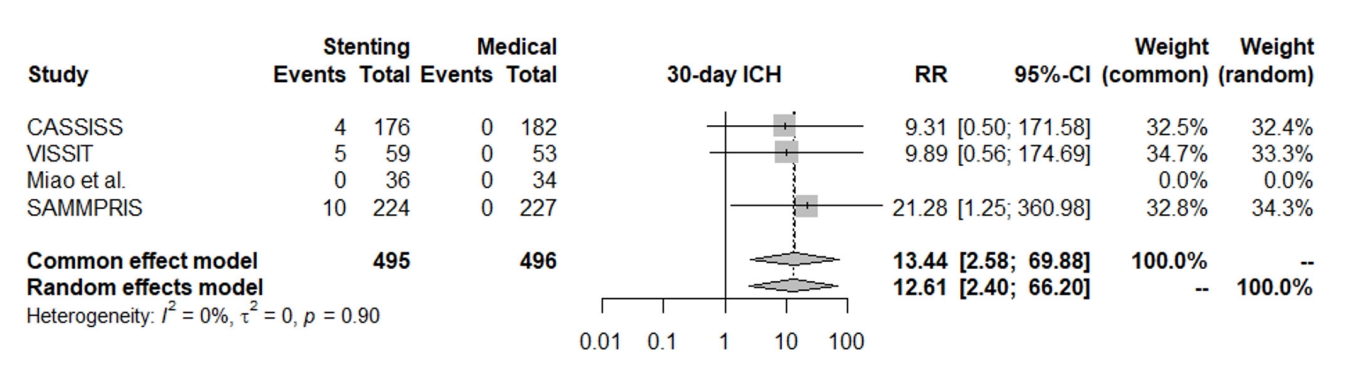
Fig. 4.
Forest plot for one-year stroke in territory or death. RR, risk ratio; CI, confidence intervals; CASSISS, China Angioplasty and Stenting for Symptomatic Intracranial Severe Stenosis; VISSIT, Vitesse Intracranial Stent Study for Ischemic Stroke Therapy; SAMMPRIS, Stenting vs. Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis
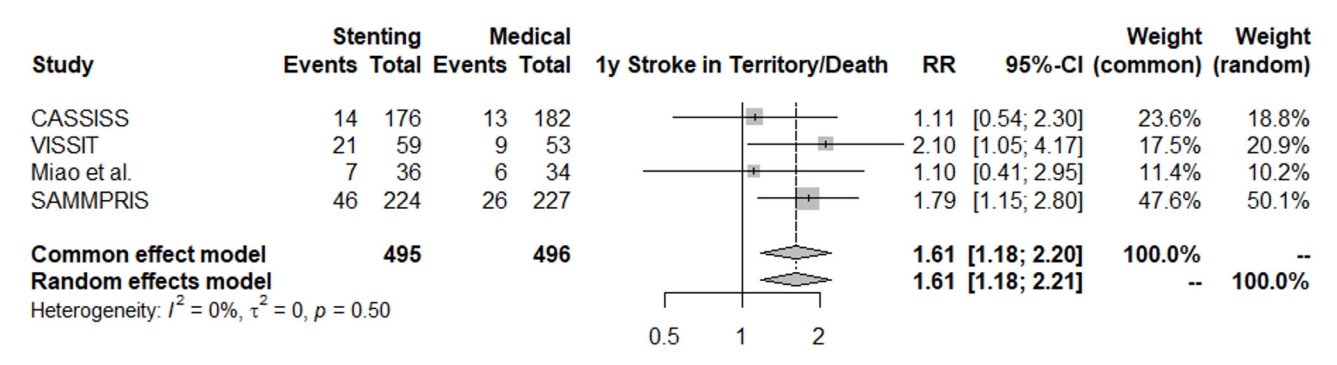
Fig. 5.
Forest plot for stroke in territory at last follow-up. RR, risk ratio; CI, confidence intervals; VISSIT, Vitesse Intracranial Stent Study for Ischemic Stroke Therapy; SAMMPRIS, Stenting vs. Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis; CASSISS, China Angioplasty and Stenting for Symptomatic Intracranial Severe Stenosis
Fig. 6.
Forest plot for disabling stroke at the last follow-up. RR, risk ratio; CI, confidence intervals; CASSISS, China Angioplasty and Stenting for Symptomatic Intracranial Severe Stenosis; VISSIT, Vitesse Intracranial Stent Study for Ischemic Stroke Therapy; SAMMPRIS, Stenting vs. Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis
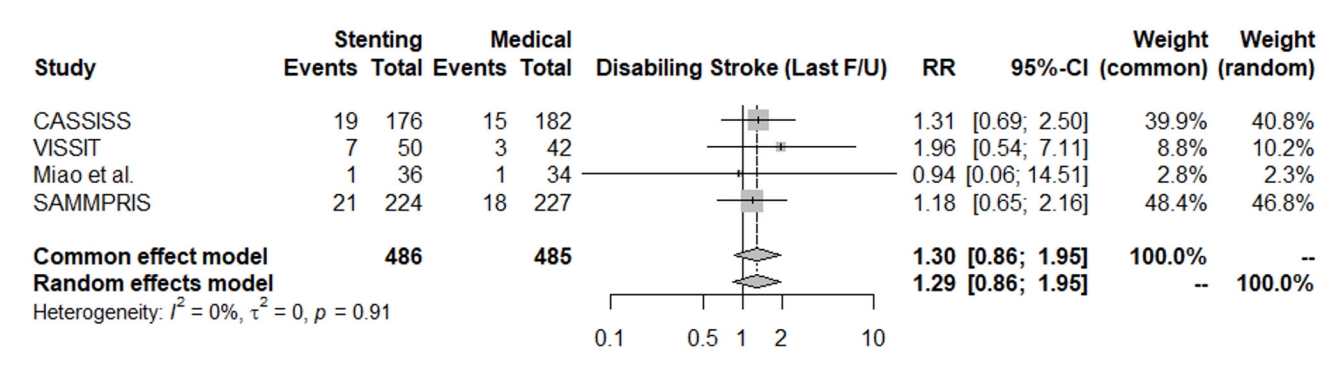
Fig. 7.
Forest plot for death at the last follow-up. RR, risk ratio; CI, confidence intervals; CASSISS, China Angioplasty and Stenting for Symptomatic Intracranial Severe Stenosis; VISSIT, Vitesse Intracranial Stent Study for Ischemic Stroke Therapy; SAMMPRIS, Stenting vs. Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis
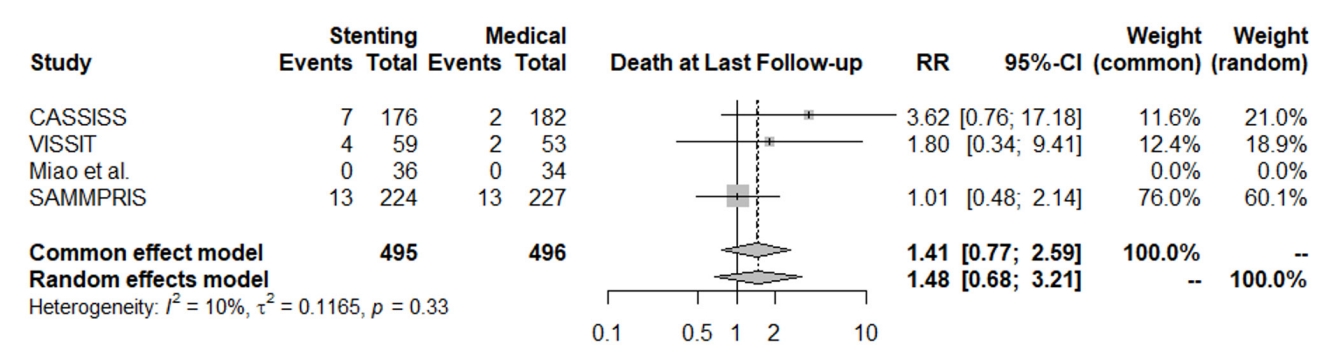
Table 1.
Baseline characteristics of included studies
CASSISS, China Angioplasty and Stenting for Symptomatic Intracranial Severe Stenosis; TIA, transient ischemic attack; VISSIT, Vitesse Intracranial Stent Study for Ischemic Stroke Therapy; RCT, randomized controlled trial; SAMMPRIS, Stenting vs. Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis
Table 2.
Studies extraction results
REFERENCES
1. Abou-Chebl A. Intracranial stenting with Wingspan: Still awaiting a safe landing. Stroke. 2011 Jul;42(7):1809-11.


2. Alexander MJ, Zauner A, Chaloupka JC, Baxter B, Callison RC, Gupta R, et al. WEAVE trial: Final results in 152 on-label patients. Stroke. 2019 Apr;50(4):889-94.

3. Arenillas JF. Intracranial atherosclerosis: Current concepts. Stroke. 2011 Jan;42(1 Suppl):S20-3.

4. Bose A, Hartmann M, Henkes H, Liu HM, Teng MM, Szikora I, et al. A novel, self-expanding, nitinol stent in medically refractory intracranial atherosclerotic stenoses: The Wingspan study. Stroke. 2007 May;38(5):1531-7.


5. Chimowitz MI, Lynn MJ, Derdeyn CP, Turan TN, Fiorella D, Lane BF, et al. Stenting versus aggressive medical therapy for intracranial arterial stenosis. N Engl J Med. 2011 Sep;365(11):993-1003.


6. Chimowitz MI, Lynn MJ, Howlett-Smith H, Stern BJ, Hertzberg VS, Frankel MR, et al. Comparison of warfarin and aspirin for symptomatic intracranial arterial stenosis. N Engl J Med. 2005 Mar;352(13):1305-16.


7. Derdeyn CP, Chimowitz MI, Lynn MJ, Fiorella D, Turan TN, Janis LS, et al. Aggressive medical treatment with or without stenting in high-risk patients with intracranial artery stenosis (SAMMPRIS): The final results of a randomised trial. Lancet. 2014 Jan;383(9914):333-41.

8. Derdeyn CP, Fiorella D, Lynn MJ, Rumboldt Z, Cloft HJ, Gibson D, et al. Mechanisms of stroke after intracranial angioplasty and stenting in the SAMMPRIS trial. Neurosurgery. 2013 May;72(5):777-95; discussion 795.



9. Fiorella D, Levy EI, Turk AS, Albuquerque FC, Niemann DB, Aagaard-Kienitz B, et al. US multicenter experience with the wingspan stent system for the treatment of intracranial atheromatous disease: Periprocedural results. Stroke. 2007 Mar;38(3):881-7.


10. Gao P, Wang D, Zhao Z, Cai Y, Li T, Shi H, et al. Multicenter prospective trial of stent placement in patients with symptomatic high-grade intracranial stenosis. AJNR Am J Neuroradiol. 2016 Jul;37(7):1275-80.



11. GBD 2019 Stroke Collaborators. Global, regional, and national burden of stroke and its risk factors, 1990-2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021 Oct;20(10):795-820.


12. Gorelick PB, Wong KS, Bae HJ, Pandey DK. Large artery intracranial occlusive disease: A large worldwide burden but a relatively neglected frontier. Stroke. 2008 Aug;39(8):2396-9.


13. Kurre W, Berkefeld J, Brassel F, Brüning R, Eckert B, Kamek S, et al. In-hospital complication rates after stent treatment of 388 symptomatic intracranial stenoses: Results from the INTRASTENT multicentric registry. Stroke. 2010 Mar;41(3):494-8.


14. Miao Z, Zhang Y, Shuai J, Jiang C, Zhu Q, Chen K, et al. Thirty-day outcome of a multicenter registry study of stenting for symptomatic intracranial artery stenosis in China. Stroke. 2015 Oct;46(10):2822-9.


15. Nahab F, Lynn MJ, Kasner SE, Alexander MJ, Klucznik R, Zaidat OO, et al. Risk factors associated with major cerebrovascular complications after intracranial stenting. Neurology. 2009 Jun;72(23):2014-9.



16. Romano JG, Prabhakaran S, Nizam A, Feldmann E, Sangha R, Cotsonis G, et al. Infarct recurrence in intracranial atherosclerosis: Results from the MyRIAD study. J Stroke Cerebrovasc Dis. 2021 Feb;30(2):105504.


17. Turan TN, Zaidat OO, Gronseth GS, Chimowitz MI, Culebras A, Furlan AJ, et al. Stroke prevention in symptomatic large artery intracranial atherosclerosis practice advisory: Report of the AAN guideline subcommittee. Neurology. 2022 Mar;98(12):486-98.



18. Wabnitz AM, Derdeyn CP, Fiorella DJ, Lynn MJ, Cotsonis GA, Liebeskind DS, et al. Hemodynamic markers in the anterior circulation as predictors of recurrent stroke in patients with intracranial stenosis. Stroke. 2019 Jan;50(1):143-7.


19. Wang T, Luo J, Wang X, Yang K, Jadhav V, Gao P, et al. Endovascular therapy versus medical treatment for symptomatic intracranial artery stenosis. Cochrane Database Syst Rev. 2020 Aug;8(8):CD013267.


20. Wang Y, Zhao X, Liu L, Soo YO, Pu Y, Pan Y, et al. Prevalence and outcomes of symptomatic intracranial large artery stenoses and occlusions in China: the Chinese Intracranial Atherosclerosis (CICAS) Study. Stroke. 2014 Mar;45(3):663-9.

21. Boden-Albala B, Wang C, Elkind MS, Rundek T, Wright CB, et al. Ischemic stroke subtype incidence among whites, blacks, and Hispanics: the Northern Manhattan Study. Circulation. 2005 Mar;111(10):1327-31.


22. Yu SC, Leung TW, Lee KT, Wong LK. Learning curve of Wingspan stenting for intracranial atherosclerosis: Single-center experience of 95 consecutive patients. J Neurointerv Surg. 2014 Apr;6(3):212-8.


23. Yu Y, Wang T, Yang K, Zhang X, Yu SCH, Luo J, et al. Timing and outcomes of intracranial stenting in the post-SAMMPRIS era: A systematic review. Front Neurol. 2021 Feb;12:637632.



24. Zaidat OO, Fitzsimmons BF, Woodward BK, Wang Z, Killer-Oberpfalzer M, Wakhloo A, et al. Effect of a balloon-expandable intracranial stent vs medical therapy on risk of stroke in patients with symptomatic intracranial stenosis: The VISSIT randomized clinical trial. JAMA. 2015 Mar;313(12):1240-8.


25. Zaidat OO, Klucznik R, Alexander MJ, Chaloupka J, Lutsep H, Barnwell S, et al. The NIH registry on use of the Wingspan stent for symptomatic 70-99% intracranial arterial stenosis. Neurology. 2008 Apr;70(17):1518-24.


-
METRICS

-
- 0 Crossref
- 0 Scopus
- 845 View
- 29 Download
- ORCID iDs
-
Adam A. Dmytriw

https://orcid.org/0000-0003-0131-5699 - Related articles



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



