 |
 |
| J Cerebrovasc Endovasc Neurosurg > Volume 22(3); 2020 > Article |
|
Abstract
Objective
Microsurgical treatment could be a good alternative for the treatment of recurrent cerebral aneurysm after coil embolization. The purpose of this study was to present our experience of microsurgical treatment for recurrent cerebral aneurysm previously treated using coil embolization.
Methods
From June 2012 to May 2019, 34 patients consecutively received microsurgical treatment for a recurrent cerebral aneurysm previously treated using coil embolization after it ruptured.
Results
Of the 34 patients with aneurysm, 33 had the aneurysm located in the anterior circulation. The most common location was the anterior communicating artery (13 cases). Immediate radiologic outcome at coil embolization was completed (n=6), residual neck (n=26), and residual sac (n=2). The reason for microsurgical treatment included rebleeding (n=12), persistent residual sac (n=1), and recurrence on follow-up study (n=21). Rebleeding occurred within 10 days after coil embolization in 10 cases, and the other 2 were due to regrowth. In the 20 recurred and saccular aneurysms, coil compaction was present in 11 aneurysms and regrowth in 9 aneurysms. Simple neck clipping (n=29) and clipping with coil mass extraction (n=3) was possible in the saccular aneurysms. The blood blister like aneurysm (n=2) were treated using bypass and endovascular internal carotid artery trapping. In the follow-up study group after microsurgical treatment there were no severe complications due to the treatment. Age, cause of retreatment, and modified Rankin Scale before microsurgery were associated with good outcome (p<0.001).
Although there are some drawbacks to the use of endovascular techniques to treat ruptured cerebral aneurysms, including incomplete occlusion and lack of long-term durability,8)10)13) they still remain the main form of treatment. However, there is a high rate of recurrence, up to 20%, of cerebral aneurysms following endovascular treatment.19) Regardless of whether recurrence occurs after microsurgery or after endovascular treatment, making a decision on how to treat the recurrent cerebral aneurysm is challenging. In order to maximize treatment outcome, it is absolutely necessary to utilize a complementary treatment strategy for the reoccurrence of a cerebral aneurysm, whether that is endovascular treatment or microsurgery.
Changing the treatment modality may be an effective strategy when it is likely that using the same treatment would be difficult or even impossible. There are already some reports about changing treatment modality, like changing from microsurgery to coil embolization, changing from coil embolization to microsurgery, or applying hybrid treatment.2)9)14)20) Among them, microsurgical treatment, such as aneurysm neck clipping or trapping with or without bypass, could be an effective and safe approach when retreatment of a recurred aneurysm that was previously treated by endovascular treatment seems difficult. However, because microsurgical treatment policies for cerebral aneurysm vary between hospitals it is unclear how effective this strategy may be. The purpose of this study was to present our experience of microsurgical treatment for recurrent cerebral aneurysm previously treated using coil embolization.
This study was a single-center retrospective study. From June 2012 to May 2019, 1,363 consecutive patients were treated with microsurgery for cerebral aneurysm. The patients who underwent microsurgery in ruptured condition were 381. Among them, 34 patients (2.5%) had a recurrent cerebral aneurysm that had been previously treated using coil embolization when it ruptured initially. Any patient whose aneurysm treatment failed or was declared as not successful enough by endovascular treatment were excluded.
The medical records and radiographic studies of these patients were retrospectively reviewed to obtain clinical and radiographic information. This retrospective study was approved by the Institutional Review Board (No. 19-0089).
Informed consent was not obtained from the individual participants included in this study because it was a retrospective medical records research.
Clinical status was measured after rebleeding (or recurrence detection), at discharge after microsurgery, and 6 months after the surgery using the modified Rankin scale (mRS) score. The angiographic outcome was measured using the “Raymond-Roy Occlusion Classification.”15)17) In cases where the dome to neck ratio was <2 or where the neck of the aneurysm was >4 mm, the aneurysm was defined as a “wide neck aneurysm”. Recurrence of the aneurysm after coiling was classified as either regrowth of the aneurysm or coil compaction. Regrowth of the aneurysm was defined as a morphological change in the aneurysm when compared with the initial angiographic morphology of the aneurysm. Coil compaction of the aneurysm was defined as a change in the coil configuration in comparison with the initial appearance of the coil mass after coil embolization without morphological changes in the aneurysm. Neck size and height of the recurred aneurysm was measured at microsurgery. Aspect ratio was defined as the height / the neck size of the aneurysm. Any recurrent aneurysm that had a coil mass placed in the dome of the aneurysm was defined as a dome type, while any having the coil mass placed in neck of the aneurysm was defined as neck type (Fig. 1). The measurement and the classification of regrowth or coil compaction of the aneurysms were determined by two neuroradiologists (HW Jeong and JW Baek). The clinical outcome was separated into good (mRS, 0-2) and poor clinical outcome (mRS, 3-6) for statistical analysis. Factors related to clinical outcome were evaluated 6 months after microsurgery.
The majority of clipping surgeries for unruptured aneurysms were performed using a superficial temporal artery (STA)-sparing mini-pterional approach introduced by our institute.1) Standard or extended pterional approaches were used for ruptured or anatomically difficult lesions, while an interhemispheric approach was used for distal anterior cerebral artery (ACA) lesions, and a far lateral approach for any vertebral artery - posterior inferior cerebellar artery (VA-PICA) lesions.
In cases where bypass surgery was used, STA-radial artery (RA)-middle cerebral artery (MCA) short interposition bypass was used. Initially, a curvilinear incision and frontotemporal craniotomy was performed, saving the STA stump. At the same time, a radial artery harvest was performed. A Sylvian dissection was performed in order to secure the operation field. End-to-side anastomosis between RA and the superior or inferior trunk of the MCA was performed, followed by end-to-end anastomosis between RA and the STA stump.
A univariate analysis was performed with age, sex, size of aneurysm, interval between coil embolization and microsurgery, dome to neck ratio, coiling technique, cause of retreatment, mRS before microsurgery, clinical outcome, and immediate radiologic outcome as independent variables using t test, Fisher’s exact test, Mann-Whitney test, and linear by linear association test. All statistical analyses were performed using SPSS for Windows (version 26; SPSS Inc., Chicago, IL, USA). The statistical significance threshold was set at p<0.05.
Demographic data and information about the initial ruptured state are described in Table 1. Out of 34 patients, 10 were male and 24 were female. Mean age at the time of microsurgery was 59.7±11.7 years old, ranging from 37 to 82. For the majority of cases (n=33) the location of the aneurysm was the anterior circulation, with the most common location being the communicating artery (A-com; 13 cases). The rest of cases found in the anterior circulation were distributed as follows: 10 cases in the posterior communicating artery (P-com), 6 in the distal ACA, 2 blood blister like aneurysms (BBL) in the distal internal carotid artery (ICA), 1 in the ICA bifurcation, and 1 paraclinoid ICA aneurysm. Only one patient had an aneurysm located in the posterior circulation, which was found to be in the VA-PICA. An initial modified Fisher Grade (mFG) score of I was found in 4 patients, a score of II in 2 patients, a score of III in 19 patients, and a score of IV in 9 patients. An initial Hunt and Hess grade (HHG) score of I was found in 10 patients, a score of II in 10 patients, a score of III in 9 patients, a score of IV in 4 patients, and a score of V in 1 patient. The average size of the aneurysm was 6.5±4.34 mm, ranging from 2.6 to 26.5. For 32 of the aneurysms, only endovascular coil embolization had been performed. However, for the two BBL aneurysms multiple overlapping stents with coils had been performed. There were 5 procedural complications including 1 procedural rupture and 4 thromboembolic events. Immediate radiologic outcome was completed in 6 cases, residual neck in 26, and residual sac in 2 (A-com and BBL).
In a subset of 12 patients, the detection of rebleeding was the reason for microsurgical treatment. Rebleeding occurred on average 142 days±413.0 after coil embolization, ranging from 1 to 1,460 days. The median value was 5 days. It is worth mentioning that in 10/12 (83%) of the cases, rebleeding occurred within 10 days of coil embolization and no angiographic change of coil compaction was detected in the follow-up angiography. The other 2 cases, in which delayed rebleeding occurred, were both neck type recurrent aneurysms (24 mm and 3.5 mm height, respectively). In 10 of the cases, direct neck clipping was possible; however, the other 2 cases required coil extraction for clipping. Clinical outcomes 6 months post-surgery were as follows: mRS 0 in 2 patients, mRS 2 in 1 patient, mRS 3 in 2 patients, mRS 4 in 2 patients, mRS 5 in 4 patients, and mRS 6 in 1 patient. The mRS 6 patient experienced 3 aneurysm ruptures and eventually developed uncontrolled intracranial pressure.
In 1 patient, the detection of a residual sac was the reason for microsurgical treatment. We first attempted to treat the BBL using multiple overlapping stents with coils, but blood flow in the BBL was still present in the 1-week follow-up angiography. Therefore, we performed STA-RA-M2 short interposition graft bypass and endovascular ICA trapping. This patient’s clinical outcome score at 6 months was mRS 0.
In a subset of 21 patients, the reason for microsurgical treatment was aneurysm recurrence detected during the follow-up study. There were 10 regrowth cases and 11 coil compaction cases. Microsurgical treatment was performed on average 686±590.2 days after coil embolization (ranging from 30 to 2160 days; median: 480 days). There were 20 saccular aneurysms and 1 BBL. For the saccular aneurysms, the height of the recurred portion was on average 3.4 mm±1.29 (aspect ratio 1.06±0.445). 17 of the saccular aneurysms were dome type and 3 were neck type. We were able to perform clipping with or without coil mass extraction in all the saccular aneurysms. The BBL was treated using STA-RA-M2 short interposition bypass and endovascular ICA trapping. Clinical outcomes 6 months post-surgery for these patients were as follows: mRS 0 in 8 patients, mRS 1 in 12 patients, and mRS 2 in 1 patient. Even though there were 2 patients who experienced transient weakness that appeared to be due to ischemia, there were no severe complications due to the microsurgical treatment that affected mRS score.
See Table 2 for all the information about the microsurgical treatment. Overall clinical outcome for patients 6 months post-surgery was as follows: mRS 0 in 11 patients, mRS 1 in 12 patients, mRS 2 in 2 patients, mRS 3 in 2 patients, mRS 4 in 2patients, mRS 5 in 4 patients, and mRS 6 in 1 patient. Importantly, there were no incidents of rebleeding detected in any of the 34 patients who received the microsurgical treatment.
We further analyzed the factors related to good clinical outcome. Table 3 shows these results. Age, cause of retreatment, and mRS before microsurgery were all associated with good outcome (p<0.001). Although the interval between coil embolization and microsurgery was associated with good outcome, it did not reach statistical significance (p=0.086).
Case 3, a 77-year-old male patient had a ruptured partially thrombosed left distal ACA aneurysm. 7 months after coiling, rebleeding occurred and microsurgery was attempted. Thrombi mixed with coils were extracted from the aneurysm for neck clipping (Fig. 2).
Case 14, a 47-year-old female patient had a ruptured distal ICA blood blister like aneurysm. Coil packing and multiple stenting was performed. Recurrence occurred after 3 months. STA-RA-M2 short interposition bypass and endovascular ICA trapping was performed (Fig. 3).
Case 17, a 35-year-old male patient had a ruptured 4.1-mm sized A-com aneurysm. Rebleeding was identified 1 week after coil embolization. An inflow of blood to the dome of the aneurysm was observed at the microsurgery. After being clipped, the aneurysm shrank, and the inflow of blood disappeared (Fig. 4).
Case 23, a 61-year-old male patient had recurrent mirror aneurysms at both internal carotid artery bifurcations (ICAB) after coiling. Clipping of both ICAB aneurysms was performed via right pterional approach. There was severe adhesion between the right ICAB aneurysm and the surrounding tissue. Therefore, partial extraction of the coil mass was needed (Fig. 5).
In this series of cases, microsurgery was performed in various situations of cerebral aneurysm recurrence after coil embolization, such as incomplete status, coil compaction, or aneurysm regrowth, irrespective of whether those were accompanied by rebleeding or not. Furthermore, various techniques were employed, including simple clip placement, coil extraction, and bypass technique. The literature emphasizes the importance of microsurgery as a retreatment option.2)6)7)16)18)20)21) Given that microsurgery requires a high surgical skill level and a careful strategy, it is mainly utilized for complicated cases.11) In fact, relatively high morbidity rates, up to 10%, have been reported previously, despite the extensive experience of the professionals.2)16)18)21) Therefore, careful preparation and planning is necessary in order to successfully treat recurrent cerebral aneurysms after coil embolization.
This study focused on patients who had a recurrent cerebral aneurysm previously treated with coil embolization after the initial rupture. The differences with respect to previous reports are the enrollment of ruptured cerebral aneurysms only, and the high proportion (12 out of 34; 35.3%) of rebleeding events leading to microsurgery. Additionally, the group that experienced rebleeding had a high proportion (10 out of 12; 83.3%) of early rebleeding (within 14 days after coil embolization). Studies show that ruptured cerebral aneurysms have different characteristics compared to unruptured ones,3) including an especially significant risk of early rebleeding after coil embolization.4)5)10)
When compared with the immediate radiologic findings obtained during coil embolization, none of the 10 early rebleeding aneurysm cases showed any change in the follow-up angiography in this study. Therefore, retreatment using endovascular techniques did not seem a viable option and instead microsurgical treatment was selected to treat the rebleeding. Importantly, all microsurgical treatments were carried out without significant complications. In our experience, it appeared that the previously implanted coils had not become organized yet and thus the artery had maintained its elasticity. We believe that this is why clipping was possible even though initially there did not seem to be enough space to place the clip. Fig. 2 provides evidence to support this theory. All considered, we believe that microsurgery represents a good treatment option in cases where a patient experiences early rebleeding after coil embolization of a previously ruptured cerebral aneurysm and where there is not enough space to pack additional coils.
In a subset of 22 cases, recurrence of the patients’ aneurysm was detected on a follow-up study and treated in an unruptured state, which makes this group uniquely different to the early rebleeding group. For this type of recurrent cerebral aneurysm, simple neck clipping is the preferred treatment option after coil embolization, but this could prove difficult in many cases. This is because endovascular implants, like coil and stent, can act as an obstacle by becoming organized and extruding into the subarachnoid space, eventually adhering to surrounding brain tissue and vessels. Therefore, the aneurysm has a large coil mass and a low height as a result, increasing the likelihood of causing damage to surrounding brain tissue, of causing unexpected intraoperative rupture, and of inadequate clipping during operation.2)16)18)21) Nevertheless, we were able to overcome these limitations and achieved a good outcome in all cases through careful patient selection and proper dissection around the aneurysm and the parent artery. We believe that the dome type recurrence might be easier to treat than the neck type. However, the neck type recurred aneurysm seems to be easier than expected to successfully clip with a penetrating clip. In our study, the saccular aneurysms that recurred on follow-up study group had ample space to place a clip, an aspect ratio of about 1.06 (mean height 3.4 mm), and no giant aneurysm. We suspect that these characteristics had a large impact on our overall positive outcome and success rate in this study.
Our study also included two cases of recurrent BBL that responded well to a STA-RA-MCA short interposition graft bypass treatment. However, treatment of BBL is still a big challenge in the field and discussion about the best treatment option is ongoing.12) Even though the reliable treatment options for BBL may vary from hospital to hospital, using the bypass technique employed in our study may prove efficacious in the treatment of recurrent BBL after endovascular treatment. In addition to BBLs, complicated and unclippable aneurysms are increasing in frequency with the development of endovascular treatment devices, such as stent and flow diverter. Therefore, the bypass technique has been essential to treating recurrent aneurysms after coil embolization failiures.11) However, more drastic surgery options will be required in these situations and we should be thoroughly prepared for it.
Previous studies have reported size of the aneurysm, posterior circulation location, intraoperative rupture, and coil mass excision as risk factors.16)18)21) Furthermore, adequate neck remnant for clip placement has been considered essential.16)18)20)21) In this study, age, rupture, and neurologic status at microsurgery were related with clinical outcome. Furthermore, the interval between coil embolization and microsurgery was longer in the good clinical outcome group. While this is not consistent with the results of previous studies, the inconsistency is likely due to the large number of early rebleeding patients included in our study, and to the limitations of a small study. The procedure of arachnoid dissection and coil extraction for ensuring an ample space for clip placement is delicate, and has a high potential risk of brain tissue and vessel injury. Therefore, tailored and safe arachnoid dissection is mandatory.
This report is a retrospective study of data from patients who underwent microsurgical treatment at single center and does not reflect overall coil embolization data. Therefore, there is a strong chance that selection bias is present and that protocol deviations have occurred. Furthermore, the sample size was small. Future studies that include larger cohorts and more cases through a multicenter approach are needed in order to further elucidate the findings of this study.
Fig. 1
(A) Drawing of a recurrent aneurysm that has a coil mass placed in the aneurysm dome (Dome type). Solid arrow indicates the neck. (B) Drawing of a recurrent aneurysm that has a coil mass placed in the neck of the aneurysm (Neck type). Solid arrow indicates the neck size and dotted arrow indicates the height of the aneurysm.
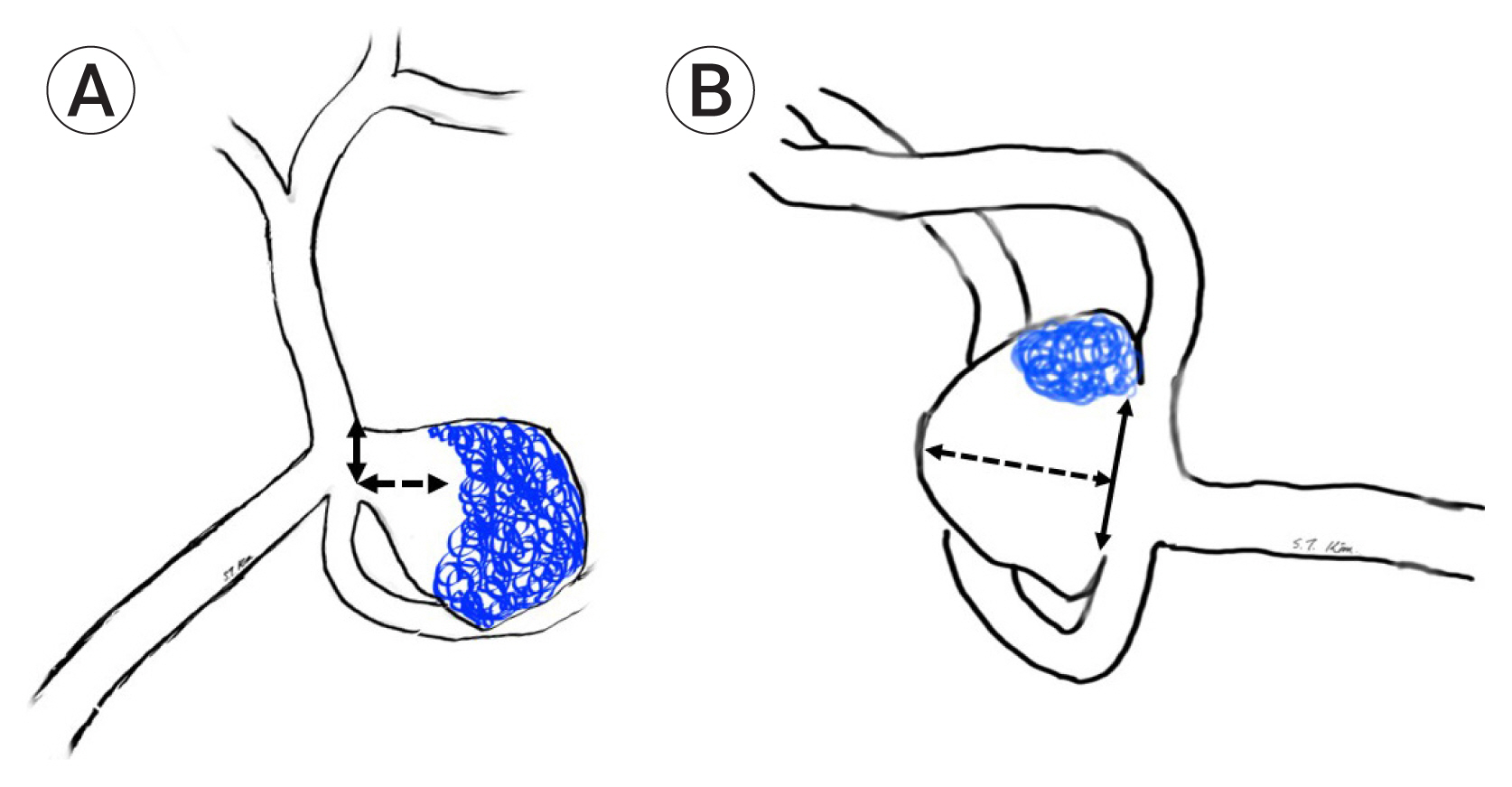
Fig. 2
Case 3, a 77-year-old male patient had a ruptured, partially thrombosed, left distal anterior cerebral artery (ACA) aneurysm. (A) A round mass was identified by a brain computed tomography (CT) scan. The mass was surrounded by subarachnoid hemorrhage and brain edema. (B) An aneurysm affecting some branching arteries of the neck was identified in left internal carotid artery (ICA) angiography; endosacular coiling was performed saving the branches at an outside hospital. (C) 7 months after coiling, rebleeding occurred. (D) Coil compaction and recanalization of the aneurysm were detected in left ICA Angiography. (E) Thrombi mixed with coils were extracted from the aneurysm. (F) Clips were securely placed, thrombi were removed, and aneurysmorrhaphy was performed. (G) Follow-up brain CT scan 1 month after microsurgery revealed reduced mass effect.
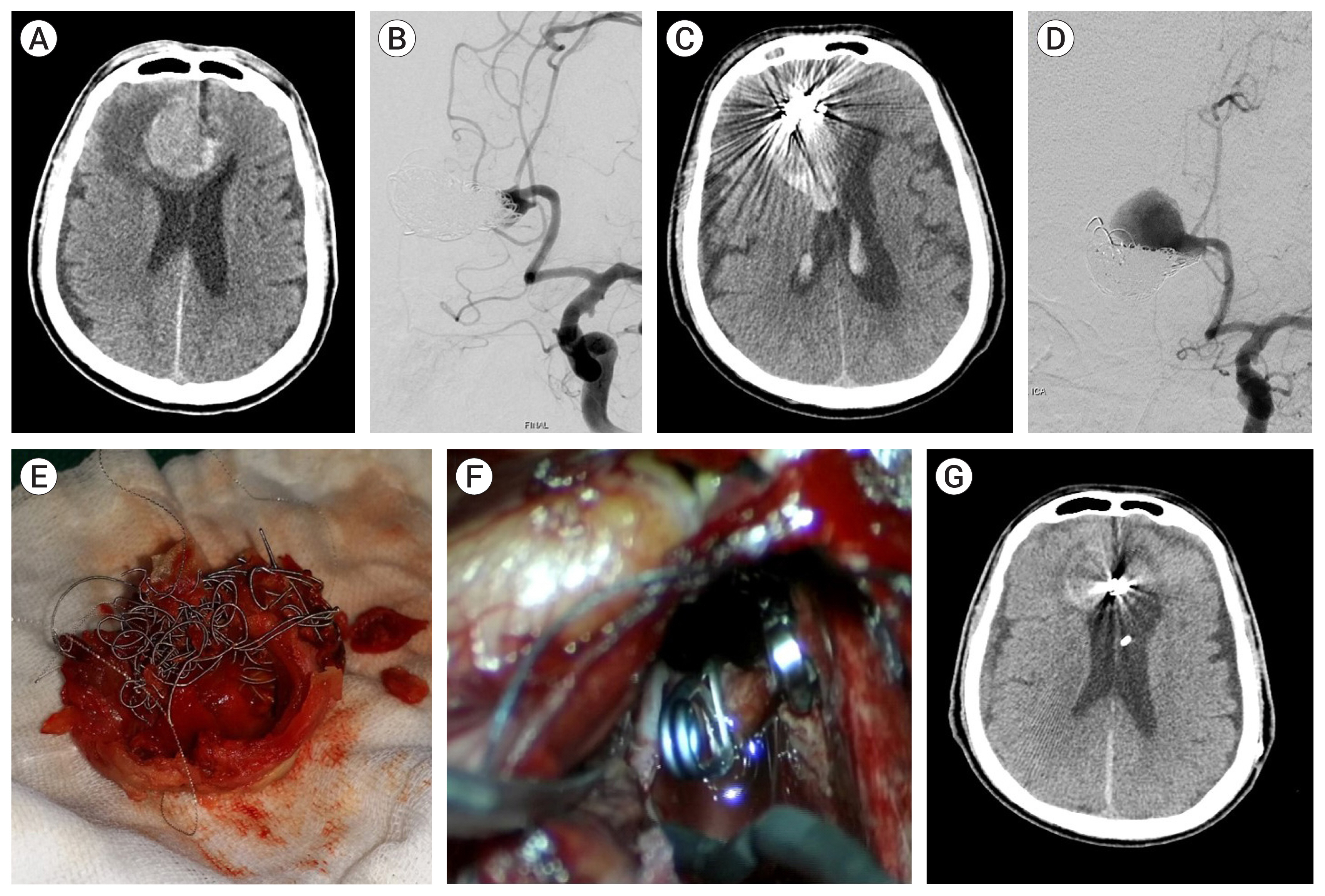
Fig. 3
Case 14, a 47-year-old female patient had a ruptured distal internal carotid artery (ICA) blood blister like aneurysm (BBL). (A) Brain computed tomography scan reveals thick subarachnoid hemorrhage around the basal cistern. (B) Angiogram reveals left distal ICA BBL aneurysm (white arrow). (C) Coil packing and multiple stenting was performed and intrasaccular thrombosis progressed well after the procedure. (D) Pathological change at the BBL was detected at follow-up angiogram after 1 month. (E) Fusiform dilatation had progressed at 3-month angiogram. (F) ICA trapping using coils promoted collateral blood flow through the anterior communicating artery and the superficial temporal artery-radial artery-M2 short interposition bypass (white dotted arrow).
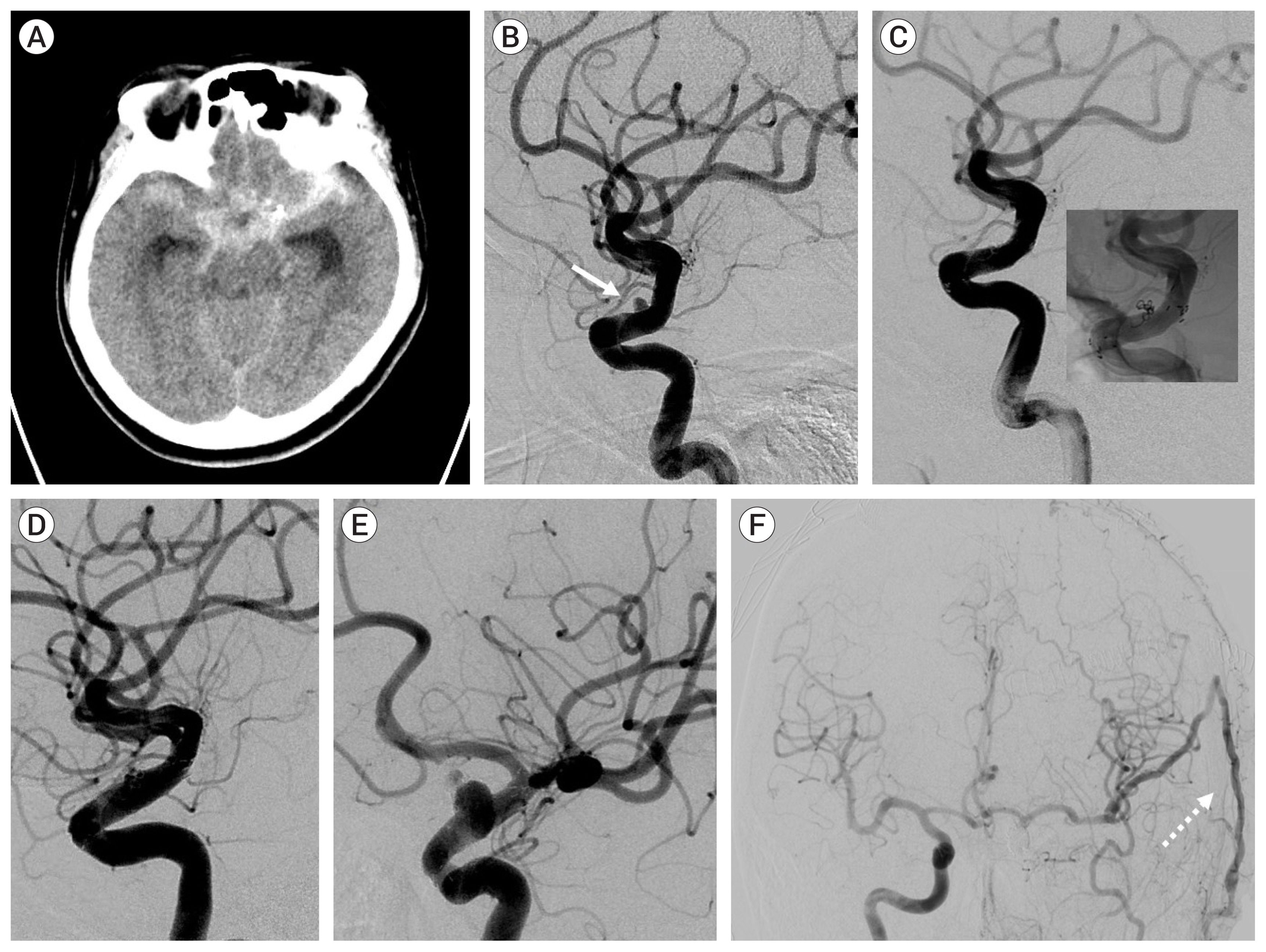
Fig. 4
Case 17, a 35-year-old male patient had a ruptured 4.1-mm sized anterior communicating artery aneurysm. (A) Rebleeding was identified by brain CT scan 1 week after coil embolization (black arrow). (B) Following rebleeding, there was no identifiable change of coil mesh (white arrow) at follow-up angiogram after rebleeding. (C) Intraoperative view of the anterior communicating artery aneurysm via the right pterional approach. We observed an inflow of blood to the dome of the aneurysm (white dotted arrow). (D) After being clipped, the aneurysm shrank, and the inflow of blood disappeared.

Fig. 5
Case 23, a 61-year-old male patient had recurring mirror aneurysms at both internal carotid artery bifurcations (ICAB) after coiling. Right side had previously ruptured. (A) Cerebral angiography revealed a neck type recurrence. (B) Clipping of both ICAB aneurysms was performed via right pterional approach. (C) At first, left ICAB aneurysm was secured by simple clipping. (D) There was severe adhesion between the right ICAB aneurysm and the surrounding tissue. Therefore, partial extraction of the coil mass partial was needed. (E) Using fenestration clip, neck clipping was possible. Unruptured posterior communicating artery aneurysm, not detected in cerebral angiography, was clipped together.
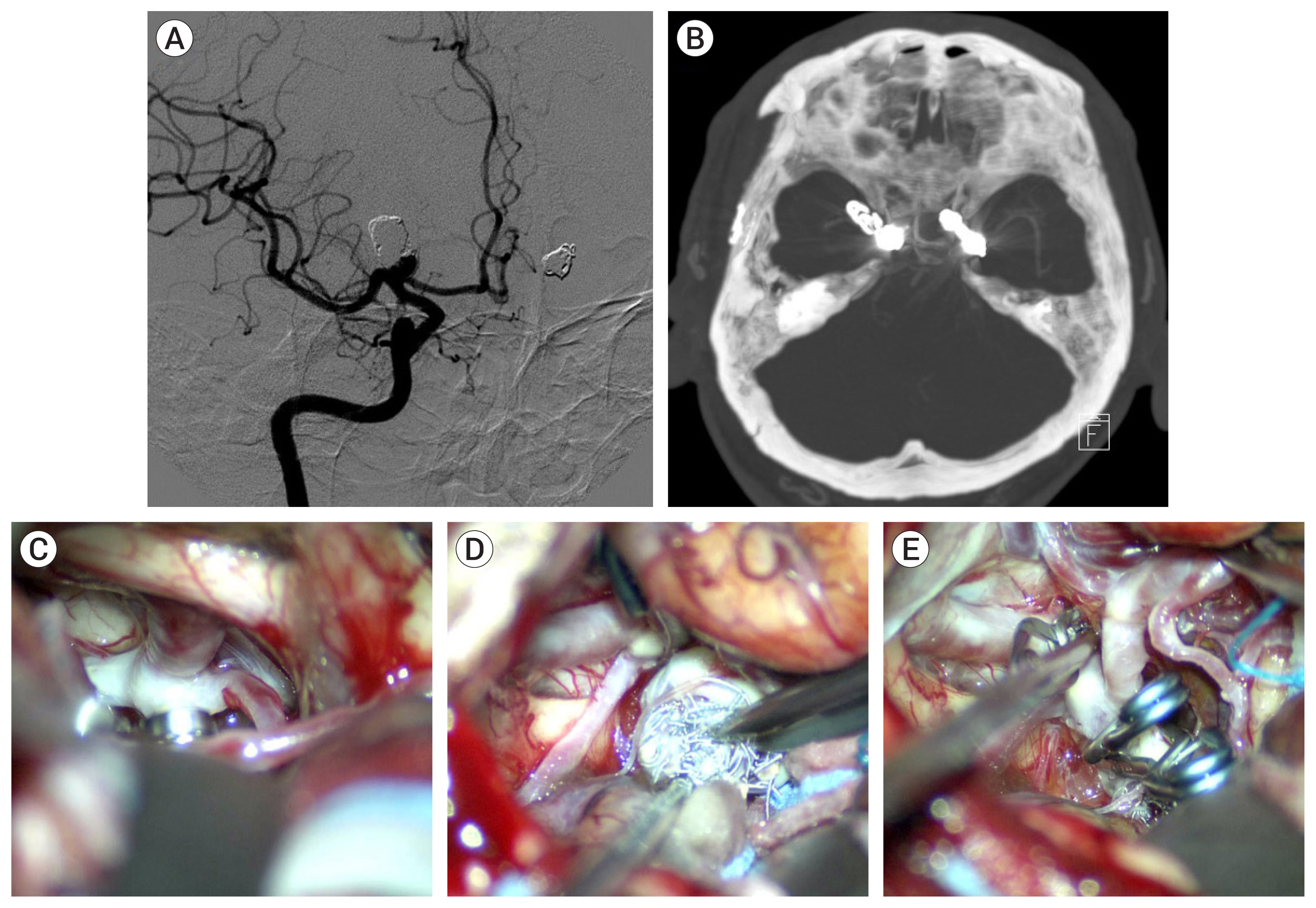
Table 1
Characteristics of patients presenting for microsurgical treatment for recurrent aneurysms after coil embolization
| Variables | Value | |
|---|---|---|
| Sex (M/F) | 10/24 | |
| Age* (Year±SD, range) | 59.7±11.7, 37 to 82 | |
| Location of aneurysm | Anterior circulation (33) | |
| A-com | 13 | |
| Distal ACA | 6 | |
| P-com | 10 | |
| ICA bifurcation | 1 | |
| ICA distal (BBL) | 2 | |
| ICA-paraclinoid | 1 | |
| Posterior circulation | ||
| VA-PICA | 1 | |
| Modified Fisher Grade | I | 4 |
| II | 2 | |
| III | 19 | |
| IV | 9 | |
|
|
||
| Hunt and Hess grade | I | 10 |
| II | 10 | |
| III | 9 | |
| IV | 4 | |
| V | 1 | |
| Size of aneurysm (mm±SD, range) | 6.5±4.34, 2.6 to 26.5 | |
| Technique of coil embolization | Simple coiling | (32) |
| Multiple stenting with coils | (2, BBLs) | |
| Procedural complications | Procedural rupture | (1) |
| Thromboembolic event | (4) | |
| Immediate radiological outcome | Total occlusion | 6 |
| Residual Neck | 26 | |
| Residual Sac | 2 (BBL, A-com) | |
Table 2
Information of clinical status, angiographic and surgical findings, and clinical outcome after microsurgical treatment
| Reason for microsurgical treatment (Total 34) | Interval between coil embolization and microsurgery | Angiographic findings at microsurgery | Surgical technique | Clinical outcome after 6 months* | |
|---|---|---|---|---|---|
| Rebleeding (12) | 142 days±413.0 | No angiographic change in 10 early rebleeding group | Direct clipping (10) | mRS | 0(2) |
| Range from 1 to 1,460 days | Coil mass extraction and clipping (2) | 2(1) | |||
| 3(2) | |||||
| Median 5 days (within 10 days, 10 aneurysms were re-ruptured) | In delayed rebleeding group (2), | 4(2) | |||
| Height 24 mm and 3.5 mm, | 5(4) | ||||
| Aspect ratio 2.4 and 1.75, | 6(1) | ||||
| Neck type (2) | |||||
|
|
|||||
| Residual sac (1, BBL) | 10 days | Blood flow in the BBL still noted | STA-RA-M2 bypass and ICA trapping using coils | mRS | (0) |
|
|
|||||
| Recur on follow-up study (21) | 686 days±86 day | Saccular aneurysm (20) | Direct clipping (19) | mRS | 0(8) |
| Range from 30 to 2,160 days | Height 3.4 mm±1.29 | Coil mass extraction and clipping (1) | 1(12) | ||
| Regrowth (10) | Median 480 days | Aspect ratio 1.06±0.445 | 2(1) | ||
| Coil compaction (11) | Dome type (17) | ||||
| Neck type (3) | |||||
| BBL (1) | STA-RA-M2 bypass and ICA trapping using coils | ||||
| Regrowth in serial follow-up | |||||
Table 3
Factors related to good clinical outcomes in the Univariate analysis
| Variables | Group (total 34) | p value | ||
|---|---|---|---|---|
|
|
||||
| Good clinical outcome* (25) | Poor clinical outcome (9) | |||
| Sex (M/F) | 8/17 | 2/7 | .692‡ | |
| Age (Year ± SD) | 54.7±8.58 | 73.7±6.50 | .000† | |
| Aneurysm size (mm, ± SD) | 5.8±2.10 | 8.6±7.60 | .938§ | |
| Interval between coil embolization and microsurgery (day, ± SD) | 578±595.2 | 186±475.1 | .086† | |
| Dome to Neck ratio (N/W) | 6/19 | 1/8 | .644‡ | |
| Coil embolization Technique (Simple coiling/Multiple stenting) | 23/2 | 9/0 | 1.000‡ | |
| Cause of retreatment (Rebleeding/Recur on follow-up study) | 3/22 | 9/0 | .000‡ | |
| mRS before microsurgery | 0 (15) | 12 | 0 | .000|| |
| (In case of rebleeding, the status at the microsurgery) | 1 (10) | 8 | 2 | |
| 2 (2) | 2 | 0 | ||
| 3 (3) | 1 | 2 | ||
| 4 (4) | 1 | 3 | ||
| 5 (3) | 1 | 2 | ||
| Immediate radiological outcome at coil embolization | Complete 6 | 5 | 1 | .962|| |
| Residual neck 29 | 18 | 8 | ||
| Residual sac 2 | 2 | 0 | ||
REFERENCES
1. Ahn JY, Kim ST, Yi KC, Lee WH, Paeng SH, Jeong YG. Superficial temporal artery-sparing mini-pterional approach for cerebral aneurysm surgery. J Korean Neurosurg Soc. 2017 Jan;60(1):8-14.



2. Arnaout OM, El Ahmadieh TY, Zammar SG, El Tecle NE, Hamade YJ, Aoun RJN, et al. Microsurgical treatment of previously coiled intracranial aneurysms: Systematic review of the literature. World Neurosurg. 2015 Aug;84(2):246-53.


3. Bijlenga P, Morita A, Ko NU, Mocco J, Morel S, Murayama Y, et al. Common data elements for subarachnoid hemorrhage and unruptured intracranial aneurysms: Recommendations from the working group on subject characteristics. Neurocrit Care. 2019 Jun;30(Suppl 1):20-27.



4. CARAT Investigators. Rates of delayed rebleeding from intracranial aneurysms are low after surgical and endovascular treatment. Stroke. 2006 Jun;37(6):1437-42.


5. Cho YD, Lee JY, Seo JH, Kang HS, Kim JE, Kwon OK, et al. Early recurrent hemorrhage after coil embolization in ruptured intracranial aneurysms. Neuroradiology. 2012 Jul;54(7):719-26.



6. Dorfer C, Gruber A, Standhardt H, Bavinzski G, Knosp E. Management of residual and recurrent aneurysms after initial endovascular treatment. Neurosurgery. 2012 Mar;70(3):537-53; discussion 553-4.



7. Izumo T, Matsuo T, Morofuji Y, Hiu T, Horie N, Hayashi K, et al. Microsurgical clipping for recurrent aneurysms after initial endovascular coil embolization. World Neurosurg. 2015 Feb;83(2):211-8.


8. Johnston SC, Dowd CF, Higashida RT, Lawton MT, Duckwiler GR, Gress DR, et al. Predictors of rehemorrhage after treatment of ruptured intracranial aneurysms: The cerebral aneurysm rerupture after treatment (CARAT) study. Stroke. 2008 Jan;39(1):120-5.


9. Kim ST, Baek JW, Jin SC, Park JH, Kim JS, Kim HY, et al. Coil embolization in patients with recurrent cerebral aneurysms who previously underwent surgical clipping. AJNR Am J Neuroradiol. 2019 Jan;40(1):116-21.



10. Kim ST, Baek JW, Lee WH, Lee KS, Kwon WH, Pyo S, et al. Causes of early rebleeding after coil embolization of ruptured cerebral aneurysms. Clin Neurol Neurosurg. 2018 Nov;174:108-16.


11. Lawton MT, Lang MJ. The future of open vascular neurosurgery: Perspectives on cavernous malformations, AVMs, and bypasses for complex aneurysms. J Neurosurg. 2019 May;130(5):1409-25.


12. Meling TR. What are the treatment options for blister-like aneurysms? Neurosurg Rev. 2017 Oct;40(4):587-93.



13. Molyneux AJ, Birks J, Clarke A, Sneade M, Kerr RS. The durability of endovascular coiling versus neurosurgical clipping of ruptured cerebral aneurysms: 18 year follow-up of the UK cohort of the international subarachnoid aneurysm trial (ISAT). Lancet. 2015 Feb;385(9969):691-7.



14. Murayama Y, Arakawa H, Ishibashi T, Kawamura D, Ebara M, Irie K, et al. Combined surgical and endovascular treatment of complex cerebrovascular diseases in the hybrid operating room. J Neurointerv Surg. 2013 Sep;5(5):489-93.


15. Raymond J, Guilbert F, Weill A, Georganos SA, Juravsky L, Lambert A, et al. Long-term angiographic recurrences after selective endovascular treatment of aneurysms with detachable coils. Stroke. 2003 Jun;34(6):1398-403.


16. Romani R, Lehto H, Laakso A, Horcajadas A, Kivisaari R, von und zu Fraunberg M, et al. Microsurgery for previously coiled aneurysms: Experience with 81 patients. Neurosurgery. 2011 Jan;68(1):140-53; discussion 153-4.



17. Roy D, Milot G, Raymond J. Endovascular treatment of unruptured aneurysms. Stroke. 2001 Sep;32(9):1998-2004.


18. Shtaya A, Dasgupta D, Millar J, Sparrow O, Bulters D, Duffill J. Outcomes of microsurgical clipping of recurrent aneurysms after endovascular coiling. World Neurosurg. 2018 Apr;112:e540-7.


19. Spetzler RF, McDougall CG, Zabramski JM, Albuquerque FC, Hills NK, Nakaji P, et al. Ten-year analysis of saccular aneurysms in the barrow ruptured aneurysm trial. J Neurosurg. 2020 Mar;132(3):771-6.

- TOOLS
-
METRICS

-
- 4 Crossref
- 0 Scopus
- 20,918 View
- 72 Download
- ORCID iDs
-
Sung-Tae Kim

https://orcid.org/0000-0002-3737-3850 - Related articles



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



